The Daily Dish
January 8, 2019
The 2018 Regulatory Record
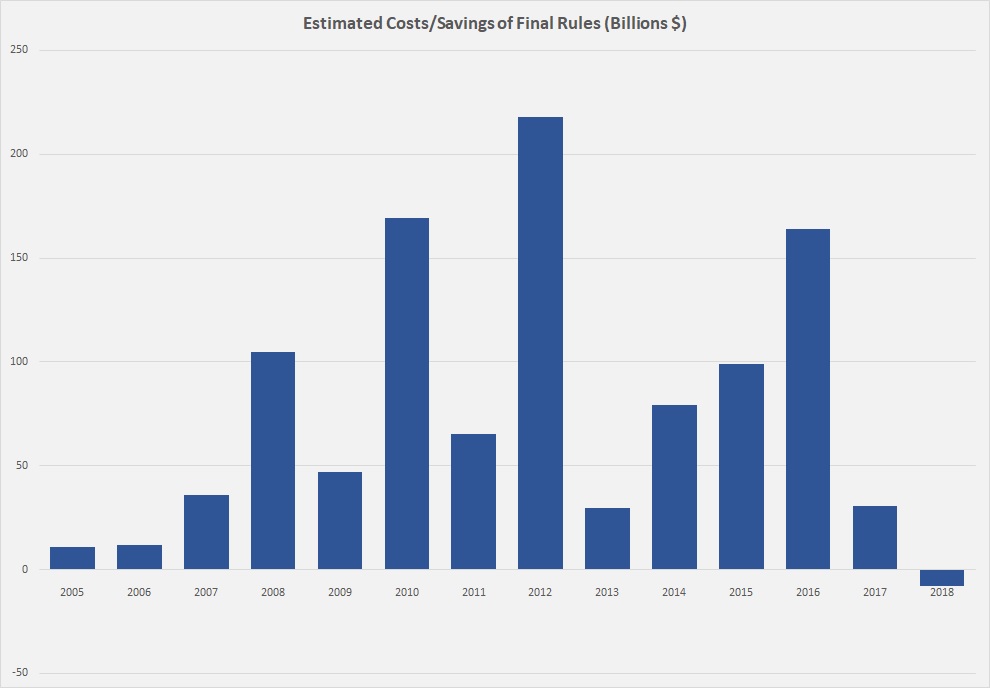
Today AAF’s Dan Bosch and Dan Goldbeck published a nice retrospective of federal regulatory activity in calendar 2018. The highlight? For the first time since 2005 — the earliest year for which AAF regulations data exist — federal agencies reduced the regulatory burden for a calendar year. (See the chart below, reproduced from their report.) By the numbers, “federal agencies finalized 324 regulations with estimated costs, savings, or paperwork impacts (the criteria for tracking on RegRodeo.com) resulting in cost savings of $7.8 billion. The agencies added 9.9 million hours of paperwork.”
Two of the top three cost-saving rules were in the Medicare program, one reason that the Department of Health and Human Services led the way with $6.0 billion in cost savings, followed by the Interior Department ($2.2 billion) and the Transportation Department ($2.0 billion). As a whole, 2018 was an extraordinary year in regulation; it is hard to single out a particular day or rule as the most exciting.
I am already sure, however, of my favorite day for 2019 (and everybody should have a favorite regulatory day): January 3! On that day, the daily Federal Register fit on one (1!) page. Hats off to Dan Goldbeck for catching this momentous event. That single page did not contain a single rule. Instead it presented two “notices,” or regulatory administrative announcements. One was about the terms of Small Business Administration disaster loans for recent storms in Illinois. The second focused on an exhibition of “Egypt: The Time of Pharaohs” in Cincinnati. It just doesn’t get any better than that.
Of course, the paucity of regulatory activity stemmed from the fact that part of the government has been closed for over two weeks. But even by shutdown standards, this is a low-cost regulatory environment. Even though the current shutdown does not cover the entire government, the page count is below the lows recorded during previous shutdowns: (1) November 14-19, 1995, which had a low of 17 pages; (2) December 16, 1995-January 6, 1996, which had a low of 21 pages; and (3) October 1-17, 2013, which had a low of 5 pages.
But maybe I’m wrong, and there will be another day in the year to come that surpasses January 3 on my list of favorites. Stay tuned for continued AAF monitoring of federal regulatory activity.
Fact of the Day
The cost to develop a new medicine is estimated to be $2.9 billion.