Week in Regulation
May 15, 2023
A Mid-May Dip
In terms of regulations formally published in the Federal Register, last week was remarkably mild. There were only five rulemakings with some quantifiable economic impact and, for the first time since the end of March, total costs fell below the billion-dollar threshold. Out of those rulemakings, the highlight of the week was the Department of Energy’s (DOE) efficiency standards rule for commercial ice makers. Across all rulemakings, agencies published $372.3 million in total costs and added 152 annual paperwork burden hours.
REGULATORY TOPLINES
- Proposed Rules: 46
- Final Rules: 47
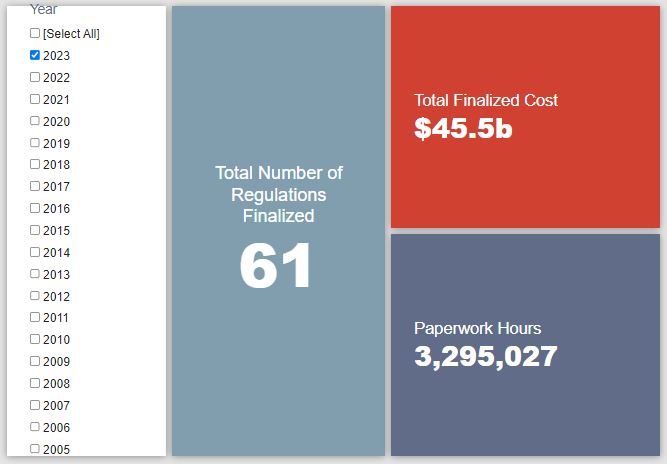
- 2023 Total Pages: 30,835
- 2023 Final Rule Costs: $45.5 billion
- 2023 Proposed Rule Costs: $285.2 billion
NOTABLE REGULATORY ACTIONS
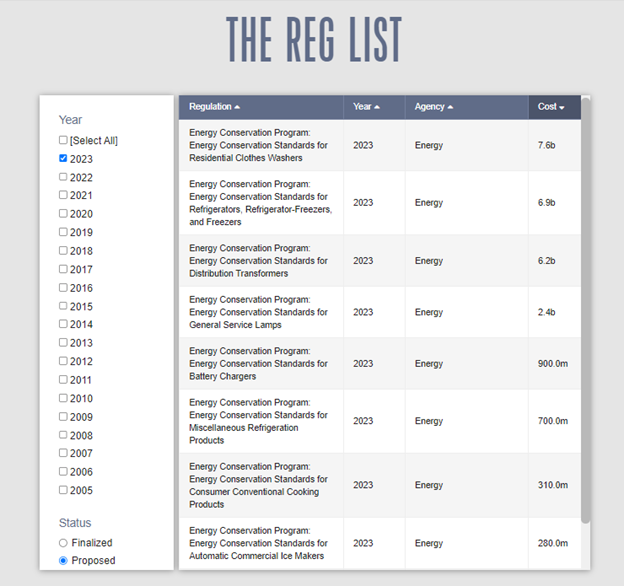
The most significant rulemaking of the week was the DOE proposal regarding “Energy Conservation Standards for Automatic Commercial Ice Makers.” This represents the first such update to the standards for these devices since a 2015 rule. DOE estimates the proposed rule will impose $280 million in total “Consumer Incremental Product Costs.” That, however, only makes it the eighth most costly set of proposed efficiency standards put forward so far this year:
TRACKING THE ADMINISTRATIONS
As we have already seen from executive orders and memos, the Biden Administration will surely provide plenty of contrasts with the Trump Administration on the regulatory front. And while there is a general expectation that the current administration will seek to broadly restore Obama-esque regulatory actions, there will also be areas where it charts its own course. Since the AAF RegRodeo data extend back to 2005, it is possible to provide weekly updates on how the top-level trends of President Biden’s regulatory record track with those of his two most recent predecessors. The following table provides the cumulative totals of final rules containing some quantified economic impact from each administration through this point in their respective terms.
![]()
There was only modest movement in the Biden Administration’s final rule cost total. Overall, it ticked up by $92 million with a Department of Housing and Urban Development rule providing the majority of that increase. The other two administrations also saw relatively quiet weeks in this respect. Trump-era costs decreased by $112 million while Obama-era costs increased by nearly $158 million. A rule on “Changes to the Medicare Claims and Medicare Prescription Drug Coverage Determination Appeals Procedures” drove the former shift and a rule concerning “Learner’s Permit Standards” for commercial drivers drove the latter.
THIS WEEK’S REGULATORY PICTURE
This week, those receiving prescriptions via telehealth appointments get a temporary reprieve.

Source: Photo by National Cancer Institute on Unsplash
Last Wednesday – the day before the formal end of the COVID-19 public health emergency (PHE) – the Drug Enforcement Administration and Department of Health and Human Services (“the agencies”) published a rule entitled “Temporary Extension of COVID-19 Telemedicine Flexibilities for Prescription of Controlled Medications.” As the title suggests, this temporary rule would allow for practitioners and patients to continue their telehealth prescription relationship beyond the end of the PHE. In particular, it provides a full extension for six months (through November 11, 2023) and then a partial extension through November 11, 2024, that only applies to practitioners and patients that have “a telemedicine relationship established via COVID–19 telemedicine prescribing flexibilities on or before November 11, 2023.”
The PHE-era standards that remain under this rule include:
-
First, the prescription must be issued for a legitimate medical purpose by a practitioner acting in the usual course of professional practice.[11]
-
Second, the prescription must be issued pursuant to a communication between a practitioner and a patient using an interactive telecommunications system referred to in 42 CFR 410.78(a)(3)—that is, audio and video equipment permitting two-way, real-time interactive communication or, for prescriptions to treat a mental health disorder—which include, but are not limited to, prescriptions for buprenorphine for opioid use disorder—a two-way, real-time audio-only communication if the distant site physician or practitioner is technically capable of using an interactive audio-video telecommunications system, but the patient is not capable of, or does not consent to, the use of video technology.[12]
-
Third, the practitioner must be authorized under their registration under 21 CFR 1301.13(e)(1)(iv) to prescribe the basic class of controlled medications specified on the prescription or exempt from obtaining a registration to dispense controlled medications under 21 U.S.C. 822(d).[13]
-
Fourth, the prescription must be consistent with all other requirements of 21 CFR part 1306.[14] [the CFR section setting general regulations for the “Prescriptions”]
This extension is one of many post-PHE policy issues that certain agencies must now reckon with. In this instance, the agencies have the stated goal of “ensuring continued access to necessary controlled medications past the COVID–PHE.” The rule is temporary in nature, however, since the agencies have a pair of proposed rules under development that seek to make more permanent changes to the regulations involved in telehealth prescriptions. Collectively, those proposed rules received more than 38,000 comments. The agencies have determined this time horizon will give them the opportunity to consider this input and develop “one or more final rules” on the issue while making sure “to disincentivize the creation of telemedicine companies that may seek to engage in problematic prescribing practices.”
TOTAL BURDENS
Since January 1, the federal government has published $330.7 billion in total net costs (with $45.5 billion in new costs from finalized rules) and 51.5 million hours of net annual paperwork burden increases (with 3.3 million hours in increases from final rules).