Week in Regulation
May 6, 2024
A Respite, of Sorts…
While the past couple of weeks were rich with milestones and superlatives, last week agencies took a bit of a breather in terms of the magnitude of rules they have published. The volume of substantive rulemaking continues apace, however – perhaps in anticipation of the Congressional Review Act “deadline” that could potentially come as soon as this month. All told, there were 25 rulemakings with some quantifiable economic impact across the week. The most significant of the bunch was a Department of Health and Human Services (HHS) rule on various hospital payment programs under its purview. Across all rulemakings, agencies published $1.4 billion in total costs and added 7.9 million annual paperwork burden hours.
REGULATORY TOPLINES
- Proposed Rules: 35
- Final Rules: 99
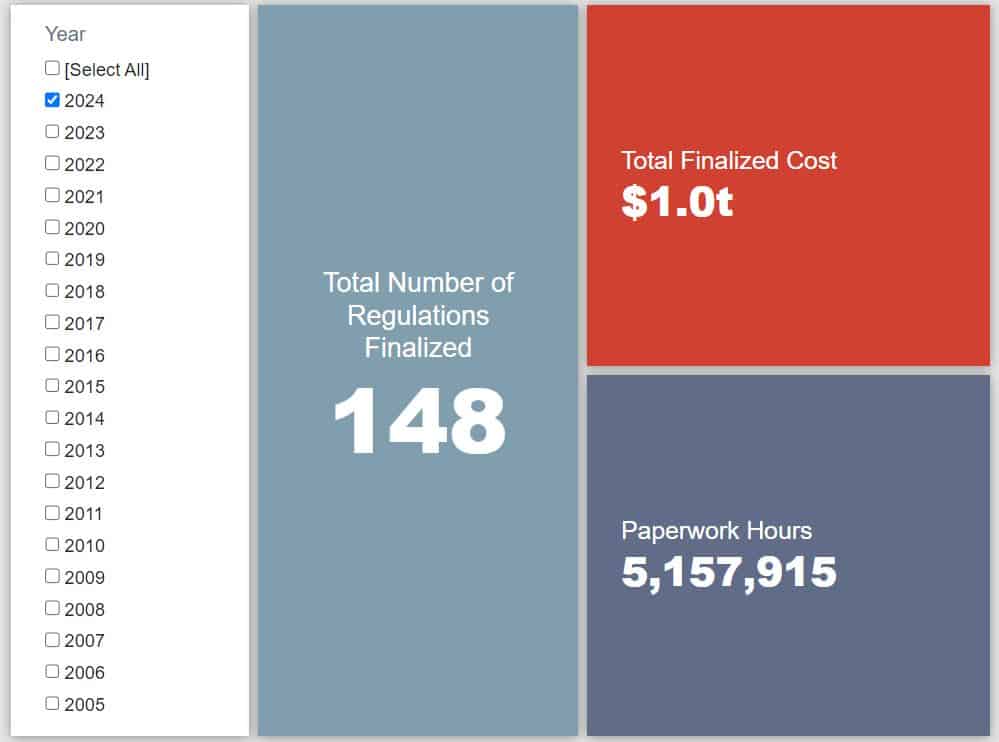
- 2024 Total Pages: 37,011
- 2024 Final Rule Costs: $1.03 trillion
- 2024 Proposed Rule Costs: $34.8 billion
NOTABLE REGULATORY ACTIONS
The most significant rulemaking of the week from a cost perspective was the HHS rule regarding “Medicare and Medicaid Programs and the Children’s Health Insurance Program [CHIP]; Hospital Inpatient Prospective Payment Systems for Acute Care Hospitals and the Long-Term Care Hospital Prospective Payment System and Policy Changes and Fiscal Year 2025 Rates; Quality Programs Requirements; and Other Policy Changes.” As the lengthy title suggests, the rule makes a series of changes to a host of payment systems under Medicare, Medicaid, and CHIP. While the rule’s main impact will be in adjusting billions of dollars in transfer payments, it will also involve updating roughly a dozen reporting requirements. These updates will result in nearly 2.7 million hours of additional paperwork and associated administrative compliance costs of roughly $205 million annually, or $614 million over a three-year period.
The other notable action of the week came as a proposed rule from the Department of Agriculture (USDA) regarding “Supplemental Nutrition Assistance Program: Program Purpose and Work Requirement Provisions of the Fiscal Responsibility Act of 2023.” Per the title, the proposal would:
Amend the Supplemental Nutrition Assistance Program (SNAP) regulations to incorporate three provisions of the Fiscal Responsibility Act of 2023 by adding to the program purpose language assisting low-income adults in obtaining employment and increasing their earnings; updating and defining the exceptions from the able-bodied adults without dependents (ABAWD) time limit; and adjusting the number of discretionary exemptions available to State agencies each year.
Much like the HHS rule discussed above, this proposed rule’s impacts come primarily on the fiscal side with $6.3 billion in reduced payments due to the work requirements and $9.2 billion in increased payments under the new ABAWD exceptions – netting out to an estimated $2.8 billion in additional outlays over the next nine years. The various changes made to the relevant programs will also involve new administrative burdens for state-level agencies and individuals. USDA estimates that such burdens will involve nearly 4.1 million hours of paperwork each year, with commensurate annual costs of roughly $64 million or $574.5 million total over the nine-year implementation window.
TRACKING THE ADMINISTRATIONS
As we have already seen from executive orders and memos, the Biden Administration will surely provide plenty of contrasts with the Trump Administration on the regulatory front. And while there is a general expectation that the current administration will seek to broadly restore Obama-esque regulatory actions, there will also be areas where it charts its own course. Since the AAF RegRodeo data extend back to 2005, it is possible to provide weekly updates on how the top-level trends of President Biden’s regulatory record track with those of his two most recent predecessors. The following table provides the cumulative totals of final rules containing some quantified economic impact from each administration through this point in their respective terms.![]() The movement in the Biden Administration’s to-date final rule tallies seems downright quaint compared to what it had been in the preceding weeks. Costs increased by $782 million while paperwork burdens jumped by more than 3.8 million hours. The HHS rule discussed above was the primary contributor to these trends. For those keeping track of how the “country” of Biden Administration Regulatory Costs stacks up in comparison to the gross domestic product of various countries, it continues to hover around Indonesia. Another rule or set of rules in the tens of billions – including, say, this $43 billion HHS rule on staffing requirements for long-term care facilities set for publication this coming week – will soon leave it chasing Spain as its next mark.
The movement in the Biden Administration’s to-date final rule tallies seems downright quaint compared to what it had been in the preceding weeks. Costs increased by $782 million while paperwork burdens jumped by more than 3.8 million hours. The HHS rule discussed above was the primary contributor to these trends. For those keeping track of how the “country” of Biden Administration Regulatory Costs stacks up in comparison to the gross domestic product of various countries, it continues to hover around Indonesia. Another rule or set of rules in the tens of billions – including, say, this $43 billion HHS rule on staffing requirements for long-term care facilities set for publication this coming week – will soon leave it chasing Spain as its next mark.
There was some notable movement in the other two administrations’ tallies, as well. The Trump Administration saw its cost total dip back well into the net-savings side of the ledger, primarily due to the nearly $200 billion in estimated cost savings from its Safer Affordable Fuel-Efficient (SAFE) Vehicles rule that sought to repeal and replace the Obama-era greenhouse gas emissions standards for passenger vehicles. That decrease in costs was somewhat blunted, however, by an HHS rule implementing aspects of the 21st Century Cures Act that involved $12 billion in net costs. The Obama Administration had the most modest week of the three administrations, with cost increases of merely $153 million and 2.4 million hours of paperwork. In what will likely come as a shock to few, an HHS measure implementing aspects of the Affordable Care Act was the most significant rulemaking in that cohort.
TOTAL BURDENS
Since January 1, the federal government has published $1.06 trillion in total net costs (with $1.03 trillion in new costs from finalized rules) and 33.4 million hours of net annual paperwork burden increases (with 5.2 million hours coming from final rules).