Weekly Checkup
May 22, 2020
An Opportunity for Telemedicine
The global COVID-19 pandemic is dramatically reshaping everything from how the House of Representatives votes on legislation to how we think about mass transit, education, and air travel. But—as is often the case with disruption—not all the implications of these changes are negative. For example, the Trump Administration has undertaken an aggressive effort to remove regulatory barriers to effective response to the pandemic, including barriers to the practice of telemedicine, potentially triggering a revolution in the delivery of health care. But this opportunity can evaporate as quickly as it appeared when the administration’s emergency actions begin to wind down.
Medicare coverage for telemedicine is mostly limited to patients who reside in designated rural areas and often requires those patients to travel to an authorized provider from where they can engage in a telehealth service. These restrictions remove much of the convenience of telemedicine. Back in March, however, the Centers for Medicare and Medicaid Services (CMS), under President Trump’s emergency declaration, undertook to expand access to telemedicine for Medicare beneficiaries.
While the president’s emergency declaration remains in effect, Medicare can reimburse for telehealth visits at the same rates as corresponding office or hospital visits, and patients can participate from their homes, without traveling to a designated facility. CMS also provided flexibility to waive cost-sharing requirements for telehealth visits paid for by federal health programs. Further, requirements that patients have a preexisting relationship with the provider from which they receive telehealth services are not being enforced for some services.
Perhaps most significantly for providers, the Department of Health and Human Services (HHS) is utilizing its enforcement discretion to waive penalties for good-faith violations of patient confidentiality regulations under the Health Insurance Portability and Accountability Act (HIPAA). As a result, providers and patients are currently able to use various free video conferencing services such as FaceTime, Skype, and Zoom.
And the regulatory changes related to telemedicine go beyond the federal level. A number of states have adjusted or suspended state laws on licensing and other restrictions that limit telemedicine. Some states are also mandating that private insurers cover telehealth on comparable terms to in-person visits, and many private insurers are voluntarily waiving copays and cost-sharing for telehealth.
The purpose behind all of these regulatory changes was to strengthen social-distancing and shelter-in-place efforts aimed at reducing infections and preventing the health care system from becoming overloaded. The effect, however, has been to show just how useful telemedicine is, and many patients and providers are hoping that this temporary telehealth flexibility might become the new norm. And they’re not alone. As AAF’s Dan Bosch wrote recently about the broader pandemic-related deregulatory effort, “the federal response has left many wondering whether the regulations that are not needed in an emergency are really necessary once the pandemic is declared over.”
Any large-scale attempt to maintain the administration’s deregulatory efforts once the public health emergency has passed will require congressional action. Further, resetting the regulatory baseline won’t be as easy as simply removing the regulations. HIPAA’s privacy protections, for example, exist for good reason, and simply waiving them permanently is hardly a desirable outcome. But perhaps it is time to revisit HIPAA and other statutes, reimbursement policies, and regulations that have constrained the practice of telemedicine. It will take thoughtful consideration, but the disruption of the pandemic has opened a door, and if policymakers seize the opportunity, our experiment in socially distanced medicine could prove to be a breakthrough for telehealth.
Chart Review: COVID-19 Test Site Distribution
Margaret Barnhorst, Heath Care Policy Intern
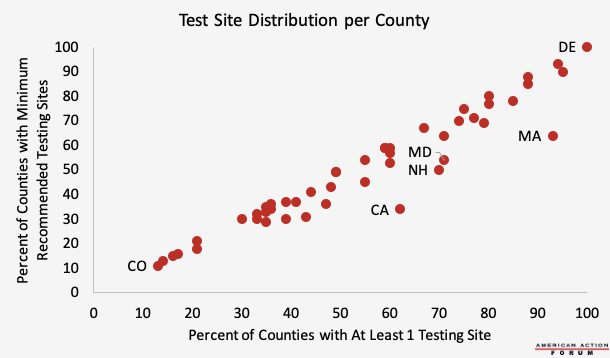
For the U.S. economy to re-open safely, experts suggest that states should have the capacity to test one percent of their population every seven days. While 46 out of 48 of the contiguous states have adequate aggregate testing capabilities to do so, test sites are distributed unevenly among counties. According to a new report from Castlight Health, nearly 54 percent of U.S. counties do not have a COVID-19 test site, including 38 percent of metro counties (those with populations greater than 50,000) and 68 percent of rural counties (those with populations less than 10,000). The presence of a testing site doesn’t mean testing is adequate in that county, however. Among the counties with at least one testing site, 58 percent still fail to meet the minimum recommended number of tests per week for their respective populations. Delaware is the only state in which all counties meet the minimum number of recommended testing sites, while states such as California and Massachusetts have far fewer counties that meet the minimum number of recommended testing sites (34 percent and 64 percent, respectively) than those that have at least one testing site (62 percent and 93 percent, respectively). To be sure, because of population variance and the distribution of COVID-19 cases, not every county necessarily needs a testing site in order to re-open safely. This analysis nonetheless helps identify inadequacies in the number of test sites, and thus testing capabilities. Opening testing sites in retail clinics with locations in widespread geographic areas, such as CVS, Walgreens, and Walmart, could help close gaps in testing and eliminate testing deserts.
Data from Castlight Health’s Local Testing Site Report
From Team Health
U.S. Dependence on Chinese Pharmaceuticals is Overstated – Director of Immigration and Trade Policy Jacqueline Varas
U.S. supply chains for pharmaceutical goods are well diversified, while domestic production of medical supplies and medications is often understated.
Health and Economy Baseline Estimates – Health Care Data Analyst Andrew Strohman
This report details the most recent updates to the Center for Health and Economy’s baseline estimates of insurance coverage, plan choice, premiums, and the federal budget.
Worth a Look
Reuters: Human trials of British coronavirus vaccine to reach 10,000
The Hill: Nursing homes struggled with infection control long before COVID-19: watchdog