Weekly Checkup
March 22, 2019
Laboratories of Drug Pricing Policy
In the ongoing policy debate about the relative value and cost associated with prescription pharmaceuticals, states are emerging as some of the most daring (and possibly reckless) policymakers.
Yesterday, Axios’ Caitlin Owens wrote about one state-driven initiative to tamp down on prices for high-cost pharmaceutical medications. A number of states, including Maryland, are looking at an approach in which they would regulate how much insurers operating in their states are allowed to pay for certain high-cost drugs. Maryland previously passed a law seeking to limit the sale price of drugs set by the manufacturer, but it was overturned by the Supreme Court as a violation of the commerce clause. It is not clear whether shifting the cap to payments made by insurers within the state—rather than prices charged by manufactures—will pass muster with the Court. In any case, the new approach illustrates how aggressive states are willing to be in trying to reduce their drug expenditures.
A more targeted and creative solution is being pursued by the state of Louisiana. Initially, in 2017, Louisiana Health Secretary Rebekah Gee advocated for the federal government to employ “government patent use” authority to seize the patents to hepatitis C cures such as Sovaldi and provide the drug at a low cost to Medicaid and prison populations whose medical costs are covered by state and federal governments. When Sovaldi first came to market, the list price was over $90,000 for a full course of treatment lasting 12 weeks. Many states struggled to absorb the costs to their Medicaid and prison populations. Both insurers and states took steps to control spending on the drug by limiting who could take it. But recent research indicates there are significant benefits to treating patients early with curative treatments like Sovaldi and its newer competitors, both in terms of reduced short- and long-term health care costs and in improved health outcomes.
As tempting as it might be for some policymakers to strip companies of their patents in an attempt to reduce drug prices and plug budget deficits, there is real long-term danger to the drug pipeline if companies believe they will be unable to make back their R&D costs. Louisiana has since changed course. In January, Governor John Bel Edwards announced a new “Netflix model” whereby the state would seek to pay a flat annual fee to one of the three manufactures of hepatitis C cures in exchange for an unlimited supply of the treatment; a similar effort is being undertaken by Washington state. As of this month, all three manufacturers have submitted proposals to Louisiana, and if all goes well, a contract could take effect on July 1. Finding the right price point to make the deal worthwhile to both the state and the manufacturer could still prove tricky, but if the model works, it could prove to be a win for states, manufacturers, and–most important–patients.
Pressure on state budgets will continue to drive bold policy innovation. Some of those policy ideas will be reckless, others ingenious. Some will run afoul of federal laws and regulations, but others will be implemented. As federal policymakers look to address drug costs, state efforts can provide a map of possible solutions and ideas to be avoided.
CHART REVIEW
Tara O’Neill Hayes, Deputy Director of Health Care Policy
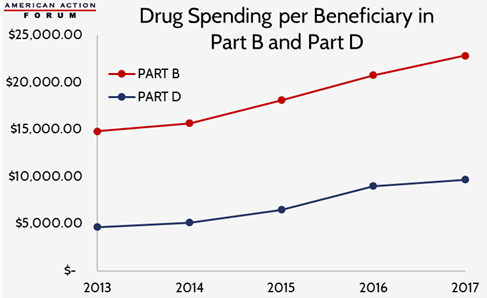
Total drug spending under the Medicare Part D prescription drug program is more than three times greater than total drug spending under Medicare Part B. There were, however, 22 times more claims for Medicare Part D drugs than Medicare Part B drugs in 2017, according to the Drug Dashboards’ data recently released by the Centers for Medicare and Medicaid Services. Of those beneficiaries actually taking drugs covered by the two programs, spending for physician-administered drugs covered under Part B has been more than two- to three-times greater over the past several years than spending on drugs covered under Part D, as shown in the chart below.
WORTH A LOOK
Washington Post: Mental health problems rise significantly among young Americans
New York Times: F.D.A Approves First Drug for Postpartum Depression
STAT News: When medicines are unaffordable, here’s how patients cut costs