Comments for the Record
June 27, 2025
Comments to USTR Regarding Foreign Nations Freeloading on American Financed Innovation
Dear Ambassador Greer:
Thank you for the opportunity to submit comments in response to the Request for Information (RFI) regarding foreign pharmaceutical pricing practices, issued pursuant to Executive Order (EO) 14297, Delivering Most-Favored-Nation Prescription Drug Pricing to American Patients (May 12, 2025). I strongly support the Office of the United States Trade Representative’s mission in opening markets throughout the world to create new opportunities and higher living standards for families, farmers, manufacturers, workers, consumers, and businesses. As the Director of Health Care Policy at the American Action Forum, I share the Office of the United States Trade Representative’s (USTR) commitment to promoting innovative, free-market solutions to build a stronger, more prosperous future. Any implementation of a most favored nation (MFN) policy or other international price-setting action is antithetical to the stated goals of USTR and the administration.
The cost of health care in the United States is a long-discussed subject in political and policy circles, with consensus that the United States pays high prices for many types of services compared to nations around the world. This is the result of many variables, including a confluence of lifestyle factors (due to an elevated American standard-of-living), aggregate wealth (due to a strong U.S. economy and hegemony), and the leading-edge innovation and care Americans receive (due to our preeminent educational and scientific systems). Based on 2023 national health expenditure data, the American consumer only spent 9.2 percent of total health care dollars on prescription drugs. This pales in comparison to hospital care, physician services, and clinical services – a combined 51.3 percent of national health expenditures. That pharmaceutical spending can be further distilled by acknowledging that 90 percent of all prescriptions in the United States are generics, which already have lower prices that international reference countries.
I write to encourage thoughtful discussion about the health care expenditure landscape, U.S. leadership in pharmaceutical innovation, and the corresponding privileged access that the United States has compared to peer countries.
When U.S. policymakers or insurers attempt to benchmark domestic prices to these suppressed foreign prices, they compound the problem and further disincentivize research and development investment in high-risk or breakthrough therapies. Simply put, importing MFN price controls, or utilizing other countries’ biased and discriminatory health assessments to assign value to life-saving pharmaceuticals – which is what many countries designated as reference points do – undermines the United States’ advantage of health care access, reduces the leverage of American wealth, and disincentives future health care leadership. While all price-setting is inefficient and should be strongly discouraged, let’s consider a few specific reasons why MFN is the wrong approach, and what can be pursued if USTR feels that there are strong, clear benefits to gain from any action on pharmaceutical pricing based on international reference countries.
OECD Is the Wrong Metric for Price Comparison
The MFN equation relies on the drug pricing data on member countries of the Organisation for Economic Co-operation and Development (OECD). But using data from OECD countries provides a poor metric: The OECD is not a wealth-based membership organization like the G-7 or the G-20. The bulk of these countries are not the most likely culprits of “free riding” on U.S. pharmaceutical innovation. In many cases, they are beneficiaries of other, non-U.S. trade agreements (such as those among European Union states) and are utilizing other trade and non-trade actions to pay for access to those pharmaceuticals. Those that are of “peer” status may have some capacity to increase their spending on health care, but the populations of those countries are not necessarily comparable to the demographics of the United States, creating further challenges in justifying drug pricing alignment.
One reason the United States is more likely to pay higher prices for drugs is simply that it has an immense amount of purchasing power, both on a per capita basis as well as on a national basis. In other words, countries with lower national wealth are less able to pay for drugs and therapies, and so correspondingly they pay less or – more likely – ration who is eligible to receive the care they can afford to provide. The implementation of an MFN pricing scheme disregards that and outsources the United States’ considerable market power to nations that centrally support their economies or have such low per capita purchasing power that practically any individual state has a higher purchasing power than they do. It is not likely the average American would appreciate tying their access to the relatively meager offerings of the pharmaceutical markets in countries that have 60 percent of the GDP of the United States, even with their comparatively lower prices.
Source: CMS Enters the MFN Fray
An oft-cited refrain is that patients in the United States are being taken advantage of, and that nations around the world are free riding on American “generosity” or “subsidies.” What this sentiment neglects to highlight is that it is better to possess a scientifically rigorous and accessible market than to be dependent of this market. The United States accounts for nearly 50 percent of global pharmaceutical revenue, the main source for funding new drug development. The U.S. drug industry invested $102 billion in pharmaceutical R&D in 2021, and over half of global R&D efforts are conducted by U.S. firms, originating the intellectual property on roughly 90 percent of all new medicines globally. Government price controls could reduce private-sector incentives to develop new treatments. A 2019 study by the University of Chicago estimated that drug price controls could reduce global R&D spending by up to $2 trillion, or a 29–60 percent reduction in R&D from 2021 to 2039, translating to 167 to 342 fewer new drug approvals during that period.
More than half of new drugs were launched first in the United States and there was an average lag of about one year between the U.S. launch and the launch in other major markets. According to a 2022 IQVIA study of the new oncology drugs launched between 2016–2020, 96 percent were available in the United States within six months of Food and Drug Administration (FDA) approval. In Canada, only 56 percent of those same drugs were available within the same period, and only 48–50 percent of new oncology drugs became available within one to two years after European Medicines Agency approval. Across all OECD countries, the United States is the clear leader in launching novel cancer therapies.
Finally, there are several mechanisms the administration envisions to enforce these goals. First, the EO mentions that the Department of Health and Human Services secretary will use rulemaking to impose an MFN price if the industry does not comply willingly with federal price-setting. There is a litany of additional options the administration is affording itself to leverage both independently and in conjunction with this rulemaking instruction, including: blanket certification that all foreign drugs are safe and that the FDA commissioner will facilitate waivers to allow their importation; legal action by the U.S. attorney general and the Federal Trade Commission chair to target companies’ anti-competitive actions; restricting exports through the Commerce secretary; and a review of drug approvals by the FDA commissioner and subsequent threat of license revocation for non-compliant companies.
Price Controls Can Undermine Pharmaceutical Innovation, Access, and Competition
Price setting in the health care industry, whether that be in the private sector or government-sponsored programs, drastically undermines pharmaceutical innovation, access, and competition.
Setting any maximum price for drugs while disregarding the intellectual property lifecycle significantly reduces the projected lifetime revenues of drugs, which affects how companies evaluate investment decisions during R&D. Pharmaceutical R&D is inherently risky, capital-intensive, and long-term; the average drug takes 10–15 years and $2–3 billion to bring to market. The Congressional Budget Office (CBO) found that pharmaceutical companies must earn a 61.8-percent rate of return on successful new drugs to see a 4.8-percent after-tax rate of return on their investment, as the failure rate for drug development is so high.
This has obvious and negative implications for the incentives to invest, particularly in areas such as oncology, rare diseases, and Alzheimer’s, where timelines are longer and scientific risk is higher. The impact is not national but rather global in scope. The United States serves as the global hub for biopharmaceutical investment, accounting for 55 percent of worldwide R&D investments and 65 percent of all development-stage funding. A recent study in Nature finds that U.S. pharmaceutical companies reinvest a disproportionately larger share of the revenue back into R&D compared to their European counterparts. For instance, U.S. companies invested 8.3 times more in R&D than they spent on marketing, compared to 1.9 times for companies headquartered in Europe.
The overall implications for innovation are significant. To see what this MFN price setting exercise may harbor in the future, let’s compare MFN to an example of price-setting quantity already in effect, the Inflation Reduction Act (IRA). An analysis by a health policy consultancy found that the IRA could reduce the number of new pharmaceutical approvals by up to 139 drugs over the next 10 years, with small- and mid-sized biotech firms being hit hardest. U.S. funding for early-stage small molecule R&D has declined by 68 percent since the IRA’s introduction, falling from $2 billion to $640 million. A study from the University of Chicago found that the IRA will lead to 135 fewer new drugs, a drop that will generate a loss of 331.5 million life years in the United States. This isn’t just a theoretical loss. While the CBO predicted only a handful of new drugs to be lost by 2039, only four months after the announcement of the IRA, at least 24 companies announced that they were curtailing their drug development.
The same dynamics affect the entry of generic competitors. Under MFN, a generic drugmaker could be ready to enter the market – including setting a launch price – when the brand name has a shift in price due to international health care dynamics. Because of the realities of generics manufacturing (slim margins, expensive active ingredients, supply chains, etc.), it is quite possible that the outcome will make the generic unprofitable. As a result, the generic may not launch at all, driving down competition in the market (making it more difficult to lower drug prices – see below). This kind of uncertainty interferes with generic entry and the healthy competition that ensures access at a reasonable price.
Outsourcing Cost and Value Assessments Is Inefficient and Dangerous
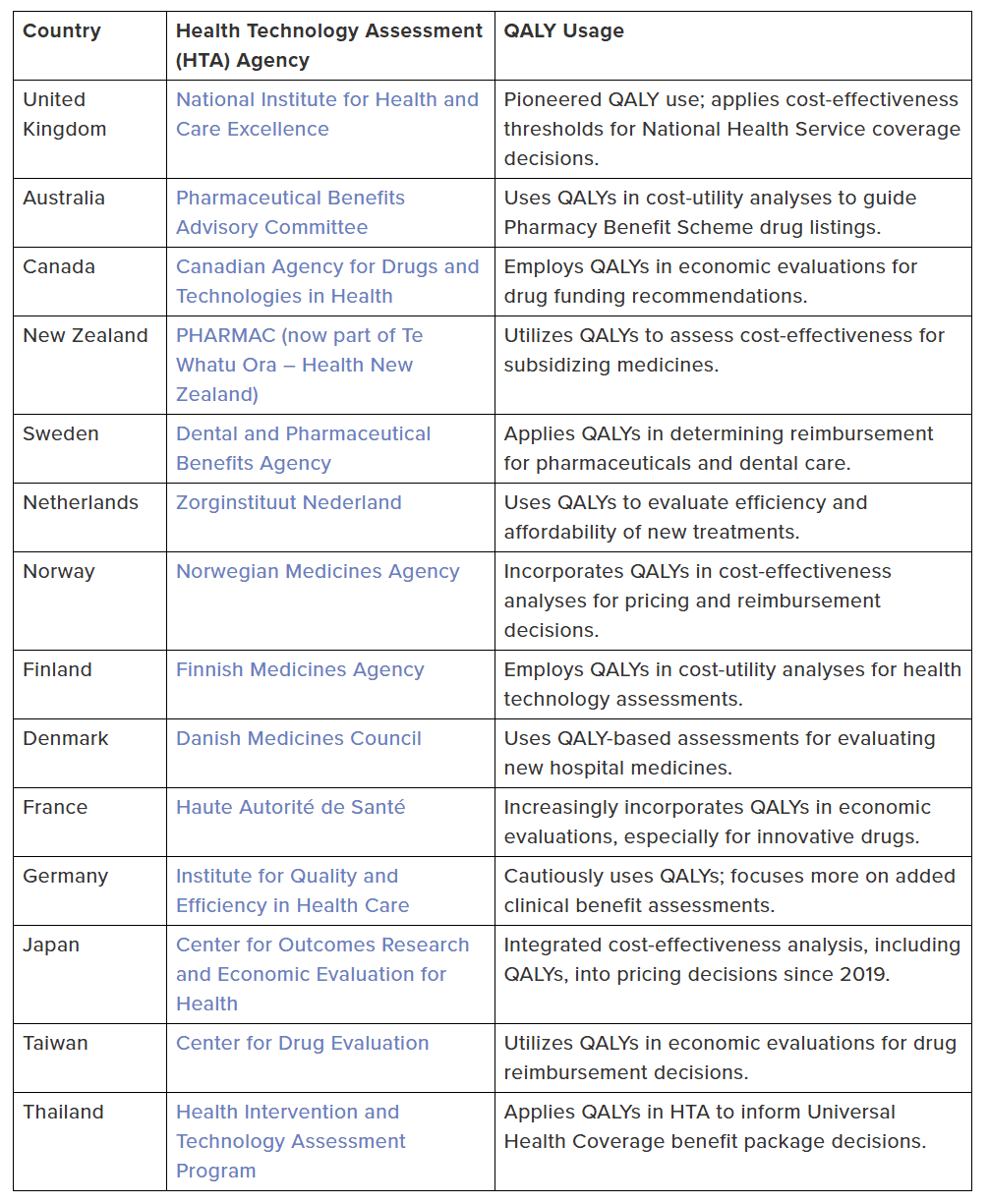
The cost, value, and efficacy of medical interventions are key components of evaluating health care outcomes across the world. In the United States, access and choice are equally important, as well. In countries that embrace single-payer health care systems, however, the cost, value, and efficacy variables are considered the more important aspects of care. This is because – with a finite budget – care must be rationed in single-payer systems to balance the demand for services with limited supply. Quality-adjusted Life Years (QALY), a metric developed in the 1970s, attempts to integrate quantity of life and quality of life into a single factor. QALYs are commonly computed as outcomes for health policy analysis and cost-effectiveness analysis to facilitate treatment and value comparisons across diseases and populations.
If desired, QALYs can then be used for decisions about the allocation of health care resources. One year in perfect health is worth 1.0 QALY. Years lived with an illness or disability are given lower values (less than one – such as 0.5 or 0.3), depending on the severity of the ailment. These scores are then used to assess the “value for money” of treatments, often through cost-per-QALY thresholds.
QALYs attempt to balance treatment investment with quality of life, but they reduce the benefit of a medical intervention to how many years of “quality” life it delivers for the cost. They raise a host of ethical questions, including how “quality of life” is measured. Because of the subjectivity in this type of model, there is inherent discrimination against older people, as any intervention will necessarily deliver fewer additional “life years,” and against those with disabilities or chronic conditions, as their “quality of life” is certain to be judged lower. This means treatments that offer more QALYs per dollar spent are more likely to be covered, while those that fall below a set threshold may be rejected – even if they benefit individual patients.
A core problem with QALYs is that they reflect population-based averages and values, not individual experiences or needs. This creates multiple challenges:
- Discrimination Against People With Disabilities and Chronic Illnesses: Because QALYs necessarily assign lower values to health states associated with disability or chronic disease, treatments for these groups often appear less “cost-effective.” This can lead to systematic underfunding of interventions for populations that already face barriers to care.
- Age and End-of-life Bias: Older adults may benefit less in QALY terms simply because they have fewer expected years of life remaining. As a result, life-extending treatments for seniors may be de-prioritized, raising troubling questions about age discrimination in public health policy.
- Neglect of Non-health Benefits: QALYs focus narrowly on physical health, omitting broader social, emotional, and economic effects of illness and treatment. Interventions that improve a caregiver’s ability to work, reduce a family’s stress, or enhance community participation are often undervalued or ignored in QALY-based evaluations.
- One-size-fits-all Measurement: Health preferences vary by culture, context, and individual experience – but QALY valuations often rely on standardized surveys or utility scores that do not reflect this diversity. The result can be an approach that oversimplifies complex medical and ethical trade-offs.
Even with the United States actively discouraging the use of QALYs, other countries regularly use them as key decision points in their health care systems.
Source: Quality-adjusted Life Years: A Single-payer Tool That Leads to Discrimination
The Most Effective Solution: Generic Competition
The USTR can have immense influence on one of the most effective ways to lower the cost of prescription drugs: the increased adoption of generics. When a brand-name drug’s patent expires, generic manufacturers can enter the market with identical formulations at significantly lower prices. This competition drives down costs for patients, payers, and other parts of the health care system.
According to an FDA study, a brand-name drug’s price begins to fall as soon as the first generic competitor is introduced. With just one generic on the market, average manufacturer prices drop by nearly 40 percent. As more competitors enter – two, four, or six – prices fall rapidly. When there are six or more generics available, prices are typically more than 95 percent lower than the original brand-name drug.
In 2022 alone, the FDA estimated that generic drug approvals saved the U.S. health care system nearly $19 billion, with more than $5 billion in savings coming from first-time generic launches. Over the long term, generics have cut costs by more than half in many markets and brought down prices by nearly 90 percent when large numbers of competitors are present.
Generics are inherently cheaper to make because they don’t carry the same development costs as branded drugs. Manufacturers can focus solely on efficient production, and each new entrant puts additional pressure on prices. The FDA ensures generics meet rigorous standards for quality and bioequivalence – giving patients access to the same therapeutic benefits as brand-name drugs at a fraction of the cost.
In a health care system increasingly concerned with affordability, generics represent a market-driven success story – delivering real value to patients without compromising safety, innovation, or access. Focusing trade efforts and market opportunities through the development, manufacturing, and usage of generics by American patients can have the largest and most sustained impact on pharmaceutical pricing.
Other Policy Opportunities to Promote Onshoring and Lower Drug Costs
On May 5, President Trump issued Executive Order 14293 that called for regulatory reform related to the construction and operationalization of pharmaceutical manufacturing facilities in the United States. I have argued that this type of reform is a much better incentive to re-shore production than tariffs or other trade barriers, including MFN.
First, let’s consider the good parts of this policy development. The EO’s emphasis on regulatory streamlining offers a more effective and sustainable alternative to tariffs as a means of encouraging domestic pharmaceutical manufacturing. While tariffs attempt to make U.S.-based production more competitive by raising the cost of imported drugs or raw materials, they may also lead to higher prices for consumers, trade retaliation, and expensive, short-term impact on supply chain issues.
In contrast, regulatory reform addresses the root causes of U.S. pharmaceutical manufacturing underinvestment: burdensome permitting processes, strict Food and Drug Administration oversight, and extended facility approval timelines. As pointed out by industry stakeholders, building a new factory can take a decade and cost about $2 billion. By accelerating environmental reviews, enhancing predictability for facility licensure, and creating inspection parity between foreign and domestic firms, the EO lowers real operational barriers to investing in U.S. manufacturing capacity.
Permitting reform is not the only obstacle to making pharmaceuticals in America: Labor costs, energy costs, and tax policy all have a role to play in building a factory. U.S. manufacturers face significantly higher labor costs than many foreign competitors. Studies show that rising labor costs have a significant negative impact on innovation and production, making workforce development programs and targeted wage subsidies essential for attracting investment. Energy-intensive pharmaceutical operations would also benefit from stable, low-cost power access – no matter the source – underscoring the need for modernized grid infrastructure and predictable energy policy. Additionally, tax incentives such as accelerated depreciation, research and development credits, or location-based manufacturing deductions could further tilt the economics in favor of U.S.-based production. Aligning these broader policies, which work in concert with international trade agreements that USTR lead engagement on, with regulatory reform may restore a globally competitive pharmaceutical manufacturing base.
Conclusion
The federal government’s expanded role in adjudicating whether a company deserves access to the U.S. market will, in effect, produce a single-payer system that determines appropriate care and outcomes for patients. A new pricing scheme that doesn’t account for U.S. purchasing power or non-monetary access privileges will dry up any return on investment that a pharmaceutical company may have previously counted on. Reduced revenue for pharmaceutical manufacturers will create downward pressure on research and development funding. This will lessen development of and access to new drugs that treat the ever-rising prevalence of chronic conditions, such as cancer and heart disease, or address rare diseases. In the past 20 years, economists have consistently found that price controls reduce the development and launch of new drugs, thus eroding American patients’ access to innovative and much-needed medicines.
Such policies were dysfunctional when enacted in President Biden’s signature Inflation Reduction Act. And they would be no less dysfunctional for an expanded MFN program to enforce the same policy for every drug in America. Implementation of any portion of this EO would set the United States more firmly on the path to a single-payer health care system than ever before.
Thank you again for the opportunity to submit comments to the USTR regarding the U.S. pharmaceutical industry and its place in the global market. I would be happy to work with you, USTR staff, and other members of the administration to promote innovative, free-market solutions and build a stronger, more prosperous future.
Sincerely,
Michael Baker
Director of Health Care Policy
American Action Forum













November 3, 2025
Comments for the Record
Employee Noncompete Agreements
Fred Ashton
Comments of Frederick C. Ashton, Jr.[1] I. Introduction and Summary The Federal Trade Commission (FTC) issued a request for information regarding employee…