Week in Regulation
April 8, 2019
Busy Week for FDA
The first week of April saw relatively high volume of activity in the pages of the Federal Register, leading to a net-deregulatory week. The Food and Drug Administration (FDA) – in perhaps a sort of swan song for now-former Commissioner Scott Gottlieb – drove most of the activity. The agency accounted for roughly one-third of all rulemakings with some quantified economic estimate and provided the bulk of the week’s cost savings. Between both proposed and final rules last week, agencies published $421.6 million in total net cost savings and added 298,433 hours of new paperwork.
REGULATORY TOPLINES
- New Proposed Rules: 57
- New Final Rules: 83
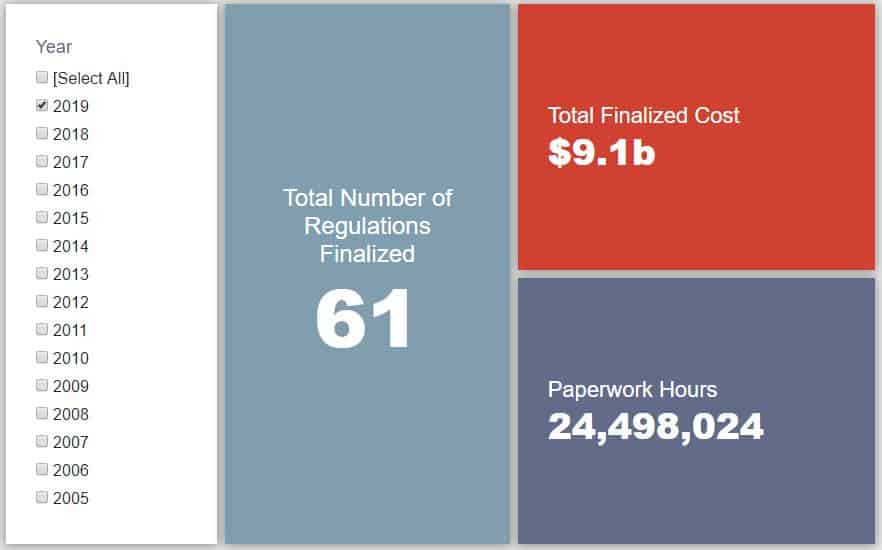
- 2019 Total Pages: 13,762
- 2019 Final Rule Costs: $9 Billion
- 2019 Proposed Rule Costs: $5.1 Billion
TRACKING THE REGULATORY BUDGET
The two most significant cost-cutting measures out of FDA were both proposed rules. The first proposal seeks to standardize and clarify the process for submitting “substantial equivalence” reports for certain tobacco products. FDA estimates that this could produce roughly $238 million in total net savings (discounted at 3 percent in perpetuity). The second measure would “amend and repeal parts of the radiological health regulations … to reduce regulatory requirements that are outdated and duplicate other means to better protect the public health against harmful exposure to radiation emitting electronic products and medical devices.” The agency estimates that this could yield $196 million in net savings. Since both rulemakings are still in the proposed stage, their cost savings do not yet apply to the fiscal year (FY) 2019 regulatory budget under Executive Order (EO) 13,771.
The FDA actions that do apply to the FY 2019 budget include one regulatory action and one deregulatory action. Both, however, were rather modest in their quantifiable scope. The first measure reclassifies “posterior cervical screw systems into class II (special controls).” This additional level of scrutiny brings half a million dollars in total costs. The second proposal reclassifies certain “in vitro diagnostic devices” in a way that reduces reporting requirements, saving roughly $729,000.
So far in FY 2019 (which began on October 1, 2018), there have been 38 deregulatory actions (per the rubric created by EO 13,771 and the administration’s subsequent guidance document) against 17 rules that increase costs and fall under the EO’s reach. Combined, these actions yield quantified net costs of roughly $10.1 billion. This total, however, includes the caveat regarding the baseline in the Department of Agriculture’s “National Bioengineered Food Disclosure Standard.” If one considers that rule to be deregulatory, the administration-wide net total is approximately $3.4 billion in net costs. The administration’s cumulative savings goal for FY 2019 is approximately $18 billion.
THIS WEEK’S REGULATORY PICTURE
One can describe “regulatory policy” in many ways: mundane, opaque, monotonous, complex, legalistic. The list goes on. In order to help provide a clearer and more straight-forward view into this world, the American Action Forum will seek to provide a brief illustration of a notable regulatory trend we have identified in a given week. This week’s entry: the final rules published by the FDA during former Commissioner Gottlieb’s tenure.
As discussed above, the bulk of regulatory activity in this week’s Federal Register came from the FDA. More than 20 percent of the combined proposed and final rules with estimated economic impacts during Gottlieb’s tenure were published this week. The likely reason for the uptick was that April 5 was Gottlieb’s final day at the agency, and the agency made clearing the deck of a few important items a priority.
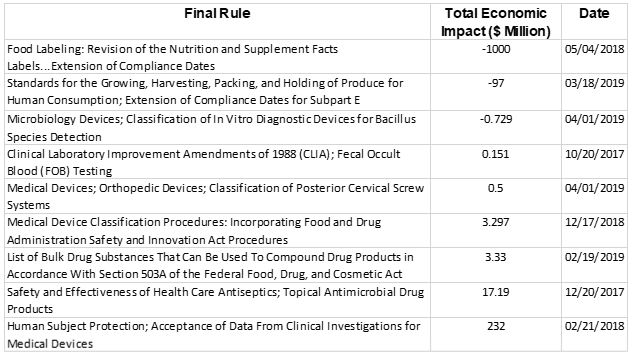
The table above shows the final rules published in the Federal Register during Gottlieb’s tenure. These rules combine to save more than $840 million. Those savings can largely be attributed to the final rule from May 2018 that extended the compliance dates for revising nutrition and supplement labels. That one action saved an estimated $1 billion. Though regulatory actions outnumber deregulatory actions by 2-to-1, they had comparatively smaller economic costs – just more than $250 million.
Yet Gottlieb will likely be most remembered for an action that isn’t reflected in the table – his efforts to limit the uptake of Electronic Nicotine Delivery Systems (ENDS) by minors by limiting what flavors are available. In May 2018, Gottlieb announced that the FDA was ending its policy of allowing ENDS manufacturer to bypass the premarket approval required for other nicotine products like cigarettes and smokeless tobacco (other than for tobacco, mint, and menthol-flavored products).
TOTAL BURDENS
Since January 1, the federal government has published $14.2 billion in net costs (with $9.1 billion in finalized costs) and 24.9 million hours of net paperwork burden increases (including roughly 24.5 million hours from final rules). Click here for the latest Reg Rodeo findings.