Week in Regulation
March 30, 2026
Substantial Health Records Rule Represents Accounting Conundrum
Last week was a lively one in the pages of the Federal Register. There were 17 rulemakings containing some kind of measurable economic impact, yet most had relatively muted effects. There was one rule from the Department of Health & Human Services (HHS), however, that stood out both in terms of its raw significance and the issues it potentially raises in arriving at a cost-benefit estimate. Overall, federal agencies published roughly $3.6 billion in total costs and added 239,011 paperwork burden hours.
REGULATORY TOPLINES
- Proposed Rules This Week: 37
- Final Rules This Week: 43
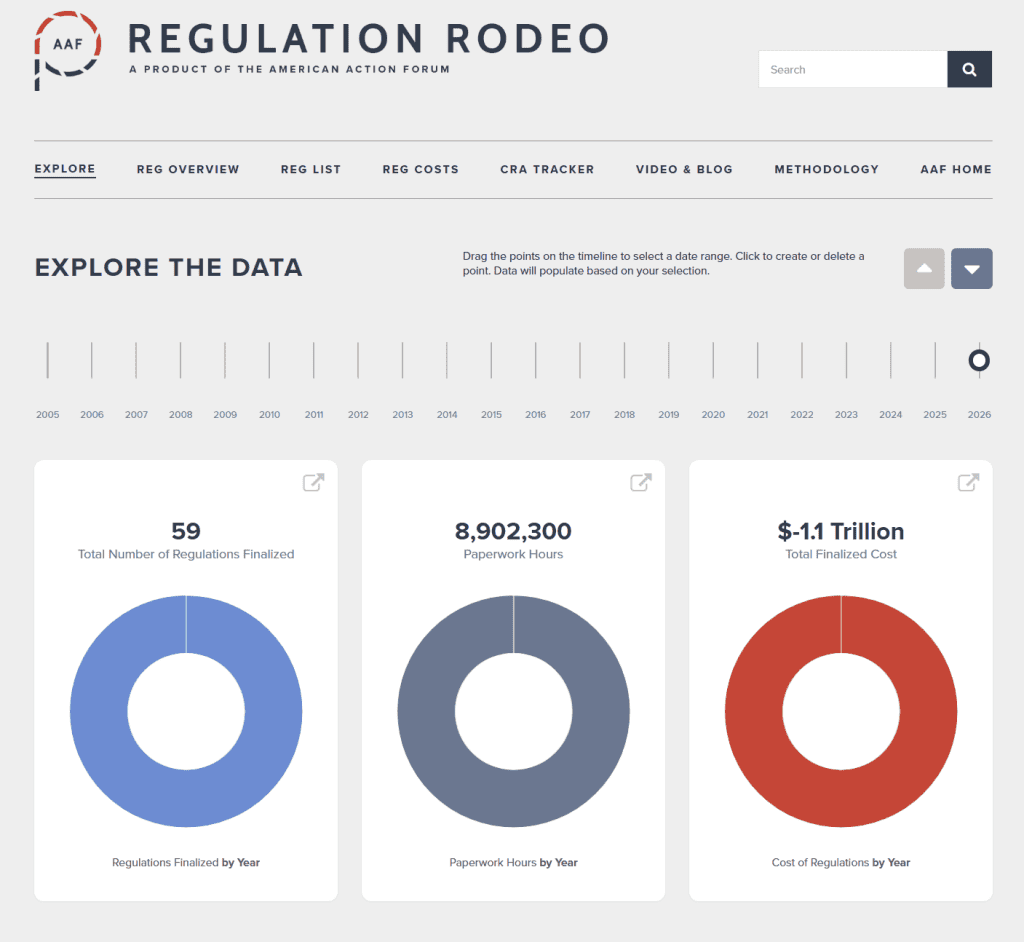
- 2026 Total Pages: 15,484
- 2026 Final Rule Costs: -$1.1 trillion
- 2026 Proposed Rule Costs: $139.3 billion
NOTABLE REGULATORY ACTIONS
The most consequential rulemaking of the week – in more ways than one – was the final rule from HHS regarding “Administrative Simplification; Adoption of Standards for Health Care Claims Attachments Transactions and Electronic Signatures.” The rule finalizes changes to “standards for the electronic exchange of clinical and administrative documentation to support claims-related processes.” The proposed rule antecedent is from back in December 2022. The magnitude of this rulemaking is sizable one way or another. The question that the rule’s current text raises is which direction its effects flow.
The rule succinctly summarizes its cost-benefit analysis as follows:
Based on the estimates included in the Regulatory Impact Analysis (RIA), the primary net annualized cost, discounted at 7 percent, to the industries is approximately $303.75 million. This estimate includes the difference between the primary net annualized costs of $478.23 million, which includes the regulatory review costs of $14.13 million, and primary net annualized savings of $781.98 million.
A careful reading of this suggests that the “net” annualized figure of $303.75 million should actually be on the savings side ($478.23 minus $781.98 million) instead of the cost side. One might assume that this was merely a typographical error, but later in the rule’s text, the agency affirms that it considers the rule a “regulatory action” for regulatory budgeting purposes.
This is all the more confounding since, in its press release announcing the rule’s publication, HHS clearly lauds its cost-saving implications. Indeed, when the American Action Forum (AAF) logged the proposed rule’s impacts in 2022, it was readily apparent that this would represent a net savings estimate. Since it appears that the agency intends to consider it a net-cost action, unless the agency provides further updates on the matter, AAF will record the rule’s estimate as expressly stated for RegRodeo and regulatory budget tracking purposes.
TRACKING TRUMP 2.0
In assessing 2026 rulemakings that include an Executive Order (EO) 14192 determination, there have been 20 “deregulatory” rules with combined total savings of $1.1 trillion against three “regulatory” rules that involve roughly $3.5 billion in costs. Adding that to the total agencies produced during 2025 (at least from rules that had a clear “regulatory” or “deregulatory” designation), the Trump Administration has enacted $1.2 trillion in total cost reductions thus far under the auspices of EO 14192. Rules for which agencies have claimed one of the EO’s explicit exemptions have accounted for an additional $508 million in costs so far in 2026.
CONGRESSIONAL REVIEW ACT (CRA)
The CRA news of this past week was much like the week before it. There was another tranche of disapproval resolutions introduced by various Democratic Senators (take, for instance, this one) seeking to reverse specific aspects of a May 2025 Consumer Financial Protection Bureau (CFPB) rule that unwound a series of that agency’s guidance documents. The rationale for this strategy remains unclear. One potential explanation may be – given that these resolutions primarily function as political messaging actions – this would give lawmakers the opportunity to take votes on individual issues rather than merely voting on the CFPB rule writ large. With 27 such resolutions of disapproval introduced thus far, there is now an expansive menu of options to choose from. It is worth remembering, however, that given current political dynamics, it remains highly unlikely that these CRA resolutions have any real chance of passing into law.
The AAF CRA tracker provides a full survey of activity under the law thus far into this term. As of today, members of the 119th Congress have introduced CRA resolutions of disapproval addressing 112 “rules” across the Biden and Trump Administrations that collectively involve $135.7 billion in estimated compliance costs. Of these, 22 have been passed into law, repealing a series of Biden Administration rules that had a combined $3 billion in associated compliance costs. The Trump Administration estimates that the repeal of this rule yields an additional $936 million in savings. While the main window of CRA action has largely passed, there are still outstanding resolutions that could move legislatively. AAF will continue to monitor and update such developments as appropriate.
TOTAL BURDENS
Since the start of 2026, the federal government has published $956.1 billion in total regulatory net cost savings (with $1.1 trillion in reductions from finalized rules) and 38.3 million hours of net annual paperwork increases (with 8.9 million hours coming from final rules).