Week in Regulation
June 21, 2022
SEC Surges Through Otherwise Quiet Week
Last week there were 10 rulemakings with some measurable economic impact. For the most part, it was looking similarly slow to the week that preceded it. Late in the week, however, a proposed rule from the Securities and Exchange Commission (SEC) turned the tide into the billion-dollar range. Across all rulemakings, agencies published $1.2 billion in total net costs and added 759,021 annual paperwork burden hours.
REGULATORY TOPLINES
- Proposed Rules: 57
- Final Rules: 55
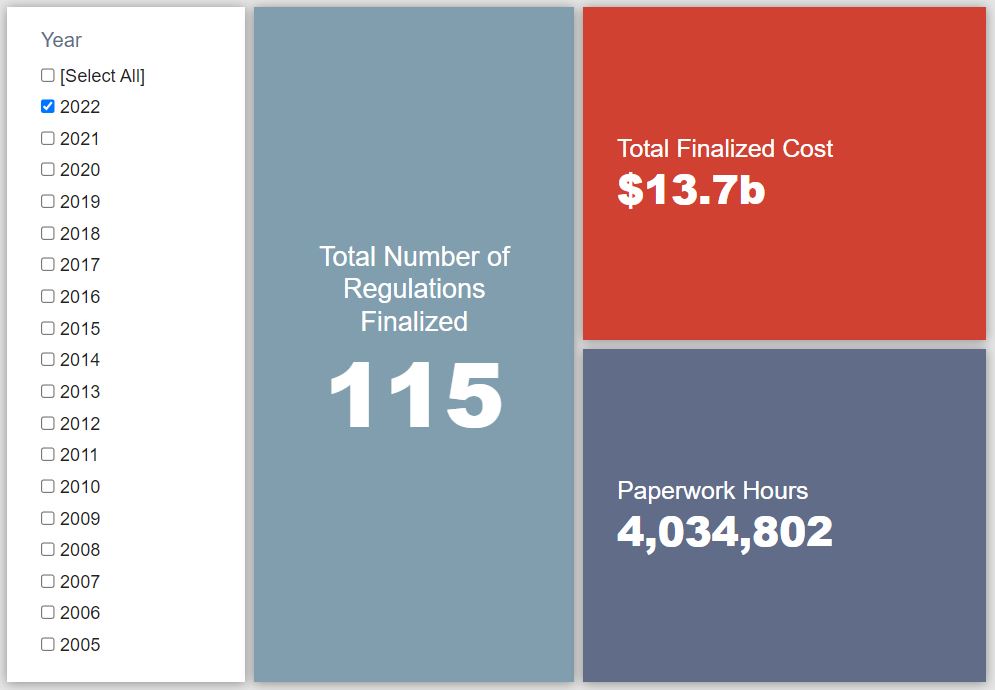
- 2022 Total Pages: 36,707
- 2022 Final Rule Costs: $13.7 billion
- 2022 Proposed Rule Costs: $76.3 billion
NOTABLE REGULATORY ACTIONS
The most significant rulemaking of the week was the SEC proposed rule regarding “Investment Company Names.” The proposal would update the regulatory code “that addresses certain broad categories of investment company names that are likely to mislead investors about an investment company’s investments and risks.” In particular, SEC seeks to tighten enforcement on various disclosure requirements related to relevant funds’ names, including “the requirement for certain funds to adopt a policy to invest at least 80% of their assets in accordance with the investment focus that the fund’s name suggests.” Per the proposal’s paperwork analysis, these additional disclosure and reporting requirements would add up to roughly 800,000 hours of paperwork and $333 million in costs annually (or $1 billion total over the three-year period in which paperwork requirements are officially approved).
TRACKING THE ADMINISTRATIONS
As we have already seen from executive orders and memos, the Biden Administration will surely provide plenty of contrasts with the Trump Administration on the regulatory front. And while there is a general expectation that the current administration will seek to broadly restore Obama-esque regulatory actions, there will also be areas where it charts its own course. Since the AAF RegRodeo data extend back to 2005, it is possible to provide weekly updates on how the top-level trends of President Biden’s regulatory record track with those of his two most recent predecessors. The following table provides the cumulative totals of final rules containing some quantified economic impact from each administration through this point in their respective terms.
![]()
Since the major item of last week came on the proposed rule side, there was minimal movement in the Biden Administration’s final rule totals. The same can be said for the Obama Administration around this time in 2010. Out of the three administrations, it was the Trump Administration that saw the most notable shift. Due primarily to an air cargo screening rule, Trump-era costs and paperwork increased by $221.6 million and 426,000 hours, respectively.
THIS WEEK’S REGULATORY PICTURE
This week, a Supreme Court decision on a Department of Health and Human Services (HHS) rule avoids directly addressing the question of deference to agencies.

On June 15, the Supreme Court released its decision in American Hospital Association v. Becerra, one of two remaining decisions from the October 2021 term that could have had broad implications on agency rulemaking. Many administrative law enthusiasts expected the case to shed light on the future of Chevron deference, a legal doctrine that defers to agencies’ interpretation of their authorizing statutes when those statutes are ambiguous. Some legal experts consider the current court likely to scrap the doctrine eventually, perhaps as soon as this term.
The case dealt with whether HHS had the ability to set varying reimbursement rates for segments of hospitals, in this case setting a lower rate for those participating in the 340B Drug Pricing Program – which caters to low-income and rural populations. Under the statute, HHS has two options to determine reimbursement rates. The first allows HHS to conduct surveys of hospitals “by hospital group.” The second allows the HHS secretary to calculate a rate. This second option does not specifically authorize HHS to set different rates by hospital group, whereas the first option does.
In 2018 and 2019, the Trump Administration set rates using the second option, but for the first time using that method set different, lower, reimbursement rates for 340B hospitals. Hospital groups challenged the rules, arguing that HHS lacked the authority to set varying rates in this manner. HHS, in response, argued that since the law did not specifically say the agency could not set varying rates using the second option, the Supreme Court should defer to HHS’s interpretation of the statute under Chevron deference.
In a unanimous decision, the Supreme Court ruled in favor of the hospital groups. Rather than addressing Chevron, however, the court instead interpreted the underlying statute as being clear in its intent on whether HHS could set varying rates using the second option. The decision concluded:
“HHS’s interpretation would make little sense given the statute’s overall structure. Under HHS’s interpretation, the agency would never need to conduct a survey of acquisition costs if it could proceed under option 2 and then do everything under option 2 that it could do under option 1. That not only would render irrelevant the survey prerequisite for varying reimbursement rates by hospital group, but also would render largely irrelevant the provision of the statute that precisely details the requirements for surveys of hospitals’ acquisition costs.”
Despite not addressing Chevron directly, the ruling does provide some indirect guidance on how the current court views the doctrine. The decision is an indication that courts should more carefully consider the statute in its full context to determine whether “ambiguous” statutes are truly vague.
TOTAL BURDENS
Since January 1, the federal government has published $90 billion in total net costs (with $13.7 billion in new costs from finalized rules) and 50.9 million hours of net annual paperwork burden increases (with 4 million hours in increases from final rules).