Weekly Checkup
May 9, 2025
Economic Policy That Actually Might Boost U.S. Drug Manufacturing
On May 5, President Trump issued an executive order (EO) that called for regulatory reform (or relief, depending on where you’re standing) related to the construction and operationalization of pharmaceutical manufacturing facilities in the United States. While I have argued that this type of reform is a much better incentive to re-shore production than tariffs, this order – which does lack some specificity in its directives – is not a panacea to the myriad issues that would still be faced by anyone who wants to build a factory.
First, let’s consider the good parts of this policy development. The EO’s emphasis on regulatory streamlining offers a more effective and sustainable alternative to tariffs as a means of encouraging domestic pharmaceutical manufacturing. While tariffs attempt to make U.S.-based production more competitive by raising the cost of imported drugs or raw materials, they may also lead to higher prices for consumers, trade retaliation, and expensive, short-term impact on supply chain issues.
In contrast, regulatory reform addresses the root causes of U.S. pharmaceutical manufacturing underinvestment: burdensome permitting processes, strict Food and Drug Administration oversight, and extended facility approval timelines. As pointed out by industry stakeholders, building a new factory can take a decade and about $2 billion. By accelerating environmental reviews, enhancing predictability for facility licensure, and creating inspection parity between foreign and domestic firms, the EO lowers real operational barriers to investing in U.S. manufacturing capacity.
Yet with these laudable goals come a few undisputed truths, including an overarching reality: When “Made in America” is the rule, things cost more. So, while onshoring manufacturing in and of itself may increase national security, it will also potentially make everything more expensive. These additional costs may be most acutely felt in the generics industry, which operates on tighter margins than branded drug manufacturers. Even if there were desire to reshore pharmaceuticals to the United States, bringing generic manufacturers that account for 90 percent of medicines used in the United States is a tall order.
Permitting reform is not the only obstacle to making pharmaceuticals in America: Labor costs, energy costs, and tax policy all have a role to play in building a factory. U.S. manufacturers face significantly higher labor costs than many foreign competitors. Studies show that rising labor costs have a significant negative impact on innovation and production, making workforce development programs and targeted wage subsidies essential for attracting investment. Energy-intensive pharmaceutical operations would also benefit from stable, low-cost power access – no matter the source – underscoring the need for modernized grid infrastructure and predictable energy policy. Additionally, tax incentives such as accelerated depreciation, research and development credits, or location-based manufacturing deductions could further tilt the economics in favor of U.S.-based production. Without aligning these broader policies, regulatory reform alone may fall short in restoring a globally competitive pharmaceutical manufacturing base.
These pains are not theoretical. Take, for example, the problems building the highly advanced TSMC computer chip manufacturing plant in Arizona – an endeavor that has been frequently cited in national security conversations. The Taiwanese company had planned a nearly $165-billion total investment in U.S.-based manufacturing. But while production at the plant started last year, the entire process was bogged down by delays and – once production started – operated at a higher cost than comparable facilities in Taiwan. Moreover, labor was both in shortage and more expensive than in other areas of the world. Material supply chains were more intensive and expensive, given the numerous regulations that govern high-tech manufacturing in the United States. Even though the plant is now functioning, the problems have made the resultant chips more expensive, with some suggesting TSMC might charge as much as 30 percent more for its Arizona-made chips than for those made in Taiwan. Pharmaceutical manufacturing is, to a great extent, comparable to the intricacies of chip manufacturing, and we should shudder to think of what the consequences might be of this type of scenario for a life-or-death drug versus a gaming chip.
While the international sections of the Trump Administration’s new EO will have the least relative impact on the eventual state of domestic manufacturing, they will have a short-term effect. As previously mentioned, the process to build, inspect, and operationalize a pharmaceutical manufacturing facility can take nearly a decade, so the interim state of the sector could have a short-term impact on the pharmaceutical trade, including price increases to account for more expensive operations (due to inspection fees and other foreign regulatory updates listed in the EO).
While the president’s recent EO is a positive step toward incentivizing pharmaceutical manufacturers to build factories and produce in the United States, there is much more that could be done. If the goal is to bring down drug prices for the American patient, tariffs should be kept permanently off the table, and actionable, economics-based policies should continue to be the order of the day.
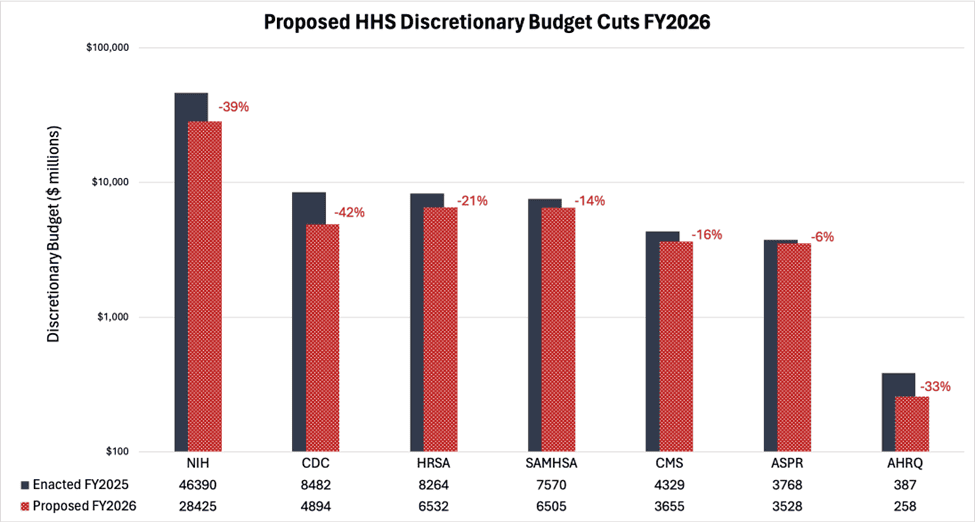
Chart Review: Significant Discretionary Budget Cuts Proposed for HHS
Nicolas Montenegro, Health Policy Intern
The president’s fiscal year (FY) 2026 “skinny budget” calls for significant cuts to and consolidation of many federal health agencies and programs, including reducing the Department of Health and Human Services’ (HHS) discretionary funding by more than $33 billion – a 26-percent reduction from the FY2025 enacted level. Importantly, the skinny budget document reflects particular priorities the administration wants to emphasize, reflecting the shifting health care priorities highlighted by the HHS secretary and others. Approximately 90 percent of HHS’ spending is mandated by spending statutes (such as Medicare and Medicaid). This and future budget documents lay the groundwork for Congress’ appropriations bills.
Seven HHS agencies and four health-oriented programs were among those deemed a priority for discretionary budget cuts in the proposal, with only negligible amounts of purported savings reallocated to fund the newly established “Make America Healthy Again” (MAHA) commission. As the chart below demonstrates, the National Institutes of Health (NIH) and the Centers for Disease Control and Prevention (CDC) were particularly targeted for spending cuts, with the president’s budget proposing reductions of about $18 billion (39 percent) and $3.6 billion (42 percent), respectively. According to the administration, the proposed reductions to these agencies would eliminate research centers that either do not align with the administration’s health care priorities or will be made redundant by new programs under the MAHA initiative. Notably, these cuts could force the elimination of funding for institutions that focus their research on chronic diseases, health care inequities, environmental health, public health preparedness and response, nursing, and others. Given the immediate pushback from members of the Senate Appropriations Committee, it is not clear to what extent Congress’ budget will reflect the president’s proposals.