Weekly Checkup
August 10, 2018
The Folly of Compulsory Licensing
Over the last few weeks there has been growing buzz around the idea of compulsory licensing in the pharmaceutical market. According to the World Trade Organization’s (WTO) definition, compulsory licensing is “when a government allows someone else to produce a patented product or process without the consent of the patent owner.” The producer pays a royalty to the patent holder, set by the government. WTO rules allow compulsory licensure but limit its scope and require various steps be undertaken before it is employed.
Compulsory licenses are largely limited to copyrighted material in the United States and exist for radio stations that want to play music and cable systems that want to broadcast over-the-air programming. In the health care industry, compulsory licensing most commonly comes into play in developing countries with antiretroviral medications; for example Zimbabwe used it on HIV drugs in 2003.
There is open debate about how effective of a tool compulsory licensing is for either reducing drug costs or increasing drug supplies. Still, researchers find that “compulsory licensing often delivered suboptimal value when compared” to alternative methods of procurement.
Some in U.S. policy circles have discussed using compulsory licensing to bring down prescription drug costs in the domestic market. Currently, 84 Democrats are cosponsoring legislation introduced by Rep. Lloyd Doggett (D-TX) that would make two key changes. First, it would repeal Medicare’s noninterference clause, (a terrible idea as detailed here, here, here and here); and second, it would authorize the Secretary of Health and Human Services to issue compulsory licenses if drug manufactures do not agree to prices the Secretary deems reasonable. In other words, the government lowers drug prices through price fixing and, if the pharmaceutical companies don’t play ball, forces them to comply anyway.
Proponents would argue that there is compensation, but the criteria on which the Secretary is supposed to base the “reasonable” licensing fee do not account for such basic factors as risk-adjusted rate of return or the administrative costs of bringing a drug to market. Even more concerning, however, is that a bill that could significantly undermine our nation’s system of intellectual property has attracted so many supporters on the left. Policymakers should take a page from what researchers know. This move is neither tenable nor effective.
Chart Review
By Jacqueline Varas, Director of Trade and Immigration Policy
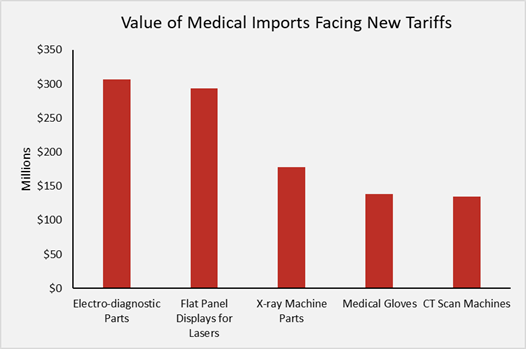
President Trump has ordered significant new tariffs over the past year, including 10 to 25 percent tariffs on over $250 billion of imports from China. These tariffs will impact roughly $1.8 billion of medical equipment imported annually, increasing nationwide prices for medical equipment by roughly $400 million per year. The most affected medical products are displayed below along with their 2017 import values.
Worth a Look
Axios: CVS Health’s rebate disclosure
Modern Healthcare: ACA subsidies cost more per person than Medicaid. Is that sustainable?
Health Affairs: ACA Marketplace Premiums Grew More Rapidly In Areas With Monopoly Insurers Than In Areas With More Competition