Weekly Checkup
January 9, 2026
When America Copies Instead of Leads
U.S. health policy keeps returning to the same rhetorical shortcut: We are an outlier compared to peer countries, so we should align with them. That framing is now doing heavy lifting in two very different debates – international reference pricing for prescription drugs and the recent move to pare back universal childhood vaccine recommendations by citing international vaccine schedules. The common thread is not simply comparison; it is policy outsourcing. When the United States uses foreign prices or foreign vaccine schedules as a decision rule, it cedes core U.S. health-economic judgments to other governments’ judgements based on their constraints and priorities. The long-run consequence is not just weaker policy; it is a retreat from American health leadership.
International comparison can be a useful diagnostic, but it is not a policy framework. “The U.S. pays more” or “the U.S. recommends more” describes an observed difference. It does not explain why the difference exists, whether the difference reflects a deliberate value choice, or whether copying another country’s arrangement could function in the U.S. health care system.
International Reference Pricing for Drugs Imports Other Countries’ Constraints
International reference pricing – often expressed through “Most-Favored-Nation” (MFN) approaches – aims to pull U.S. prices toward the lowest prices observed in selected peer nations. The political appeal is obvious: If other developed countries pay less, why shouldn’t Americans? But foreign prices are not neutral market signals. They are downstream outputs of other countries’ health technology assessment rules, global budgets, centralized negotiation structures, coverage limits, and – in many cases – explicit access management. Pegging the United States to those prices without considering the full institutional context is not “getting a better deal.” It is allowing foreign systems to set the shadow price of American innovation and access.
Three distortions tend to follow. First, negotiating leverage shifts offshore. If a foreign payer knows its price can become a reference point for the United States, it gains incremental bargaining power while the U.S. loses it. Second, launch and access incentives warp. A common concern is that reference pricing can contribute to delays in marketing new drugs (thus delaying patient access) in lower-price markets precisely because prices reverberate across borders. Third, the United States fails to build its own health care valuation.
None of this argues for complacency about U.S. drug affordability. But international benchmarks are a poor foundation for U.S. health economics. A serious strategy should be built around domestic levers – competition policy, generic and biosimilar uptake, benefit design, rebate dynamics, and targeted reforms to federal purchasing and coverage – rather than outsourcing to a foreign index.
Using International Vaccine Schedules to Justify Paring Back U.S. Childhood Recommendations Is a Category Error
The same governance error appears in the administration’s move to pare back universal childhood vaccine recommendations by referencing international vaccine schedules. Vaccine schedules are not interchangeable menus. They are built around local disease burden, transmission patterns, health system architecture, and the policy tools that determine whether risk-based approaches will succeed in practice.
Countries that recommend fewer routine childhood vaccines often have very different conditions: more uniform primary care access, stronger longitudinal patient tracking, integrated health records, and policy mechanisms that reliably bring high-risk children back into care. The United States, by contrast, has decentralized access, variable continuity, and a delivery system where friction is endemic.
Even popular claims that the United States gives dramatically more “doses” than peer countries frequently depend on methodological choices that inflate the gap – counting annual flu and COVID doses, treating combination-vaccine math differently, or comparing “maximum possible” shots rather than typical schedules. The point is not that every U.S. recommendation is beyond debate. The point is that “other countries do less and are fine” is a thin and potentially misleading premise for reshaping a prevention architecture that depends on clarity and uptake.
The Shared Failure
International reference pricing for drugs and international vaccine schedule mimicry share three governance failures.
- They treat foreign outcomes as authoritative without importing foreign inputs: You cannot import the price outcomes of centralized systems without importing their institutions. You cannot import another country’s vaccine schedule without its longitudinal primary care continuity and tracking infrastructure.
- They replace domestic deliberation with an external benchmark: In both cases, the benchmark becomes the justification, crowding out transparent U.S.-specific evidence and accountability.
- They erode U.S. leadership by normalizing dependency: The United States has long set global standards in biomedicine and prevention. When U.S. policy is justified as “matching peers” rather than grounded in first principles, the U.S. signals it is no longer leading – only following.
Policies that externalize decision-making to foreign indices risk degrading both prevention performance and innovation incentives, while weakening the credibility of U.S. health governance. Instead, the United States should learn globally but decide domestically. International evidence of best practices should inform U.S. policy, not substitute for its own interests and due diligence. A leadership-oriented posture would use global comparisons as inputs – then make decisions anchored in U.S. realities. If the United States wants to remain a leader in health care, it must act like one: set standards, build durable institutions, and openly acknowledge comparative, international tradeoffs.
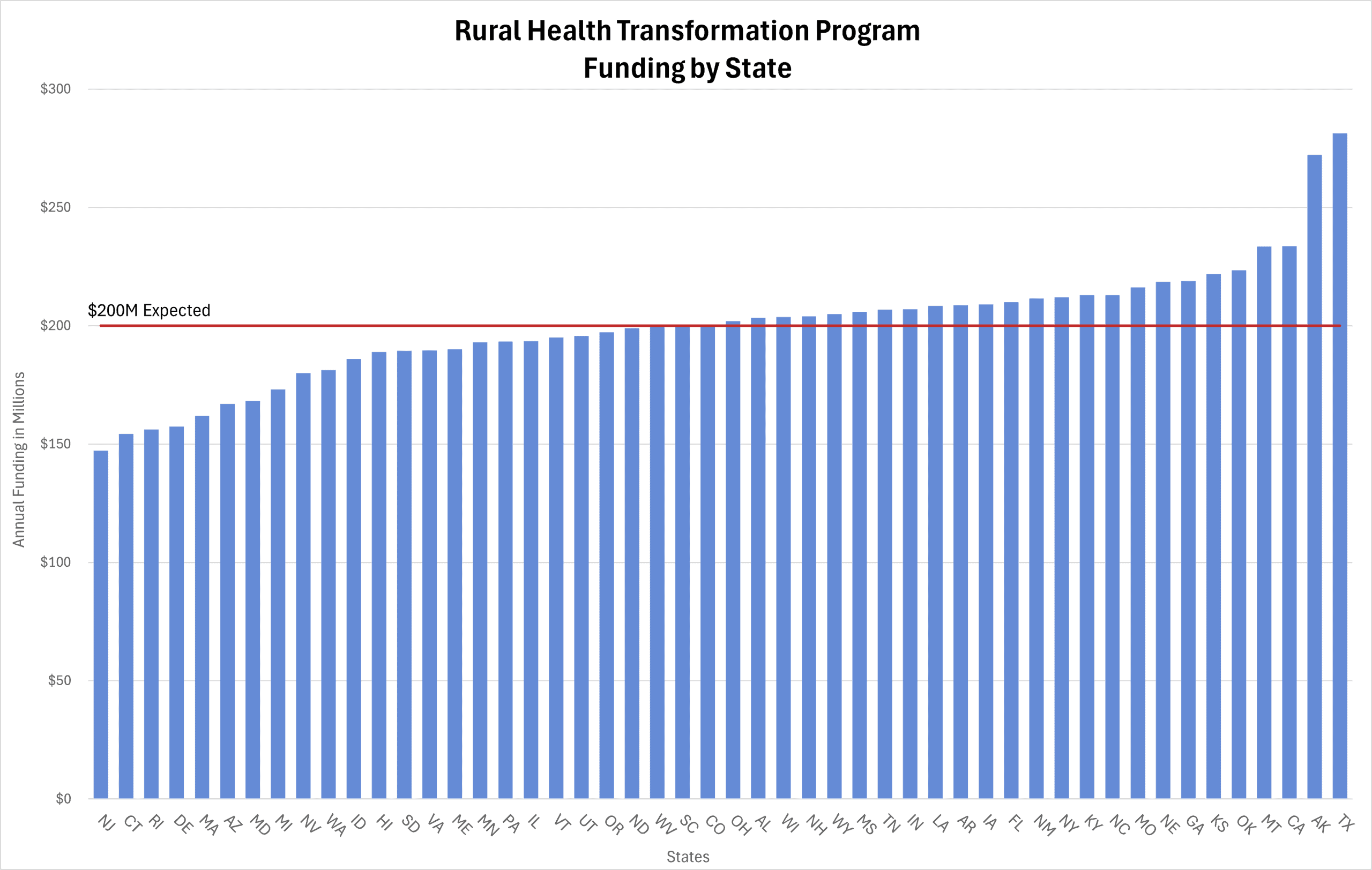
Chart Review: CMS Awards Rural Health Transformation Grants
Jack Leimann, Health Care Policy Analyst
Last week, the Centers for Medicare & Medicaid Services (CMS) awarded all 50 states a portion of the $50 billion available through the Rural Health Transformation (RHT) Program. The program, authorized by the One Big Beautiful Bill Act through 2030, is intended to improve rural communities’ health care access and quality. Per the law, CMS distributed 50 percent of the RHT funding equally among all states, and 50 percent based on other individualized factors, such as a state’s rural demographic data. CMS defines rural as “nonmetropolitan counties” identified by the Health Resources & Services Administration. Analysis of the state’s rural demographic data included the absolute size of rural population, percentage of population located in rural areas, and area of state in total square miles. As such, an interesting correlation illustrated by the chart is that the four-largest states received the most funding, and the four-smallest states received the least. For example, Alaska received the second-most funding of all states with $272 million for fiscal year (FY) 2026. This is attributed to Alaska being the largest state in total square miles, with a widely dispersed population. In comparison, New Jersey, the fourth-smallest state in total square miles, received the lowest funding despite it having the highest population per square mile.
The chart below details the RHT funding awarded to each state, beginning in FY 2026. CMS suggested states use $200 million as a reference for their annual budgets to formulate their individual proposals, represented by the “$200M Expected” line in the chart below. This number was based on every state receiving an equal amount of funding, yet as noted, this was not the case due to other variables in the distribution formula.