Weekly Checkup
May 1, 2020
The Challenges of Accelerated Vaccine Development
The global population desperately needs an effective vaccine (or a reliable therapeutic treatment) in order return to some semblance of normalcy. While some experts suggest we are at least 12 to 18 months away from an effective vaccine, Pfizer has recently announced it will begin testing a potential vaccine in the United States next week, with plans to have it available for emergency use in the fall. This announcement comes on the heels of Oxford University reporting a similar timeline for their vaccine the day prior.
Should these efforts bear fruit over the next couple of months, this is fantastic news. The successful development of a vaccine a full year ahead of the already ambitious timelines would be a testament to our biopharmaceutical sector, and we should applaud and encourage the efforts of companies and universities to continue this unprecedented pace of research and development. There are, however, a couple of challenges that come with this breakneck speed of development, and we should pause to consider them.
First, we have to adequately consider the risk of a vaccine leading to immune enhancement, i.e. aiding the virus instead of aiding the immune system. Different types of immune enhancement exist, such as antibody-dependent enhancement (ADE) or cell-based enhancements like Th2 immunopathology. Researchers have seen ADE in tissue and animal models for severe acute respiratory syndrome (SARS); the antibodies that were intended to help key the immune system to the virus instead helped the virus enter cells. Th2 immunopathology—in which an improper T-cell response triggers allergic inflammation that can damage airways—has been observed in experiments with aged mice, raising concerns about this problem translating to the elderly population who are already disproportionately affected by COVID-19. This form of immune enhancement was also seen during early vaccine development for SARS.
Fortunately, preliminary experiments on COVID-19 indicate that ADE will likely not be a problem, although more research is needed to solidify this claim. Additionally, by revising their approach and taking a bit more time to mitigate the problem of Th2 immunopathology in the SARS vaccine, researchers were able to successfully move the vaccine into clinical trials. Scientists would do well to integrate any technical knowledge that came from this hiccup as well as heed the lesson that a slightly slower schedule can lead to a better vaccine.
The second major problem is scaled production of the vaccine. As Bill Gates discussed in a recent op-ed, many of the top candidates for COVID-19 vaccines require unique equipment to manufacture. Producing these vaccines at scale is more complicated than simply shifting existing infrastructure, as manufacturers would need to build out new capacity. This will take both time and additional investment.
This problem seems much more readily addressed. As Gates noted, the private sector can mobilize much faster than the government, and he is funding the production of facilities for the top seven vaccine candidates. While it is difficult for private companies to shoulder this level of risk (apart from private philanthropy), the federal government can more readily do so, and private-public partnerships may be an effective means of distributing this risk. Further, the Trump Administration is touting Operation Warp Speed as a “Manhattan Project-Style effort” with the goal of propping up both development and production of a vaccine and making 300 million doses available by January of 2021.
While we need to attend to these problems with the seriousness they deserve, we should simultaneously cheer on private- and public-sector efforts to expedite vaccine development. Private companies will lead the charge to protect the health of the global populations, but policymakers should ensure they are doing all they can to maintain the pace of vaccine development while minimizing the risks.
Chart Review: Statistics and Narratives Around the Pandemic
Andrew Strohman, Health Care Data Analyst
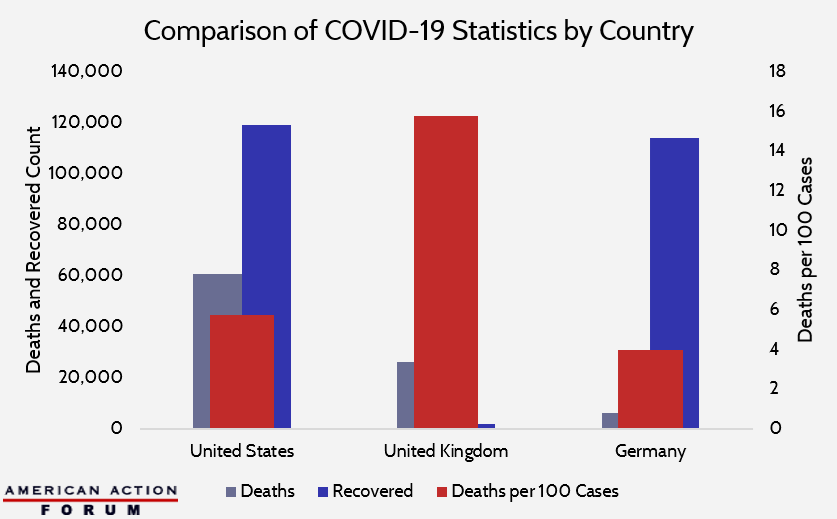
As of April 29, the United States is leading the world in coronavirus cases, with 1,056,780, and deaths, with 60,823. By comparison, the United Kingdom has 165,221 cases with 26,097 deaths, while Germany has 161,187 cases and only 6,401 deaths. These raw figures seem to indicate the United States is handling the pandemic poorly, but the focus on just these two raw counts is shortsighted. In addition to cases and deaths, the United States also leads the world in recoveries. Further, the United States has nearly five times the population of the United Kingdom and four times the population of Germany. Assuming a uniform rate of spread across these countries and adjusting for differences in populations shrinks these discrepancies dramatically. Finally, creating a simple rate of deaths per 100 cases places the United States—with a rate of approximately 5.8 deaths per 100 cases—in between the United Kingdom, with a rate of 15.8, and Germany, with a rate of 4.0. These figures undermine any narrative that the United States is failing to address the pandemic as well as other, similar countries.
Data obtained from the Centers for Disease Control and Prevention, the World Health Organization, and the European Centre for Disease Prevention and Control
From Team Health
Federal and State Actions to Address Insulin Costs – Director of Human Welfare Policy Tara O’Neill Hayes, Margaret Barnhorst, and Josee Farmer
State and federal policymakers are considering a variety of options for bringing down the cost of insulin, but most would have unintended consequences that would simply shift costs.
Avoidance, Treatment, Protection, and Tradeoffs in the Pandemic – AAF President Douglas Holtz-Eakin
Advances in the medical science of COVID-19 are important in their own right, but they also change how one thinks about the economic policy priorities.
Worth a Look
New York Times: How Coronavirus Mutates and Spreads
FierceHealthcare: CMS eases requirements for COVID-19 tests, boosts payments for telephonic visits