Research
March 3, 2020
Understanding the Insulin Market
Executive Summary
- Of the growing diabetic population in the United States, roughly 8.3 million people require insulin to regulate blood glucose levels, and it is estimated that worldwide insulin use will increase 20 percent by the year 2030.
- Competition in the insulin market has been limited for several reasons: Products are not interchangeable, there are essentially only three insulin manufacturers in the U.S. market, and no regulatory pathway has existed to allow biosimilar products to create generic-like competition for insulin.
- By the end of 2020, nearly all existing patents for insulin products will have expired and a regulatory pathway for biosimilar insulin in the United States will be available—changes that should facilitate downward price pressure.
Introduction
Diabetes is a growing health issue in America and is the seventh leading cause of death. Over 34.2 million Americans, or 10.5 percent of the U.S. population, had diabetes as of 2018.[1] Roughly 1.4 million Americans have type 1 diabetes, in which the body’s pancreas cannot produce insulin on its own, usually a result of genetic abnormalities.[2] The remaining majority of diabetics have type 2, in which the body cannot properly use the insulin it has, often because of certain lifestyle factors. According to the Centers for Disease Control and Prevention (CDC), another 84.1 million people in America have prediabetes, a condition that if left untreated will lead to type 2 diabetes within 5 years.[3] Gestational diabetes occurs in roughly 14 percent of all pregnancies in the United States and often goes away following delivery, but for nearly half of these women, it will develop into type 2 diabetes within a few years following the pregnancy.[4]
Of the growing diabetic population in America, nearly a quarter use insulin.[5] Insulin is a necessary medication for nearly all type 1 diabetics (1.4 million people), and roughly 21 percent of type 2 diabetics (6.9 million people) must also use insulin to bring their blood glucose levels down to a healthy range.[6] Without insulin, patients are likely to suffer from blurred vision, weight loss, and intolerable thirst. Eventually, uncontrolled diabetes can lead to blindness, kidney failure, gangrene, loss of limbs, and ultimately death.[7]
Insulin Manufacturers and Products
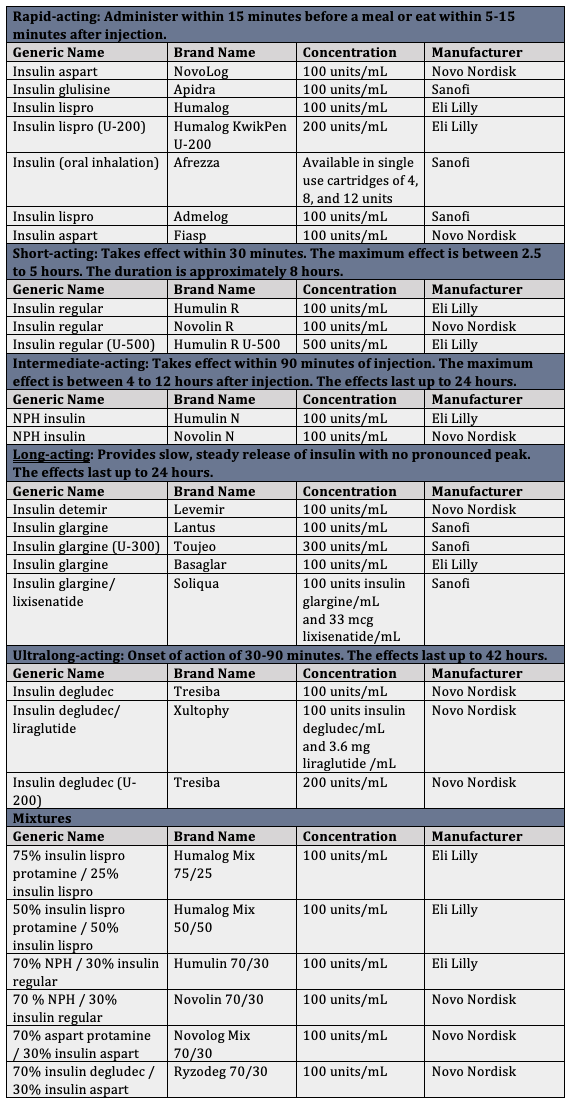
There are 3 primary insulin manufacturers in the U.S. market—Eli Lilly, Novo Nordisk, and Sanofi—that represent over 90 percent of the global insulin market and produce almost 100 percent of the insulin supply in the United States.[8] In general, each manufacturer makes multiple types of insulin, and the only monopoly held over one specific type is Novo Nordisk’s ultra-long-acting insulin.
Source: Consumer Med Safety[9]
Insulin was first extracted from animals in 1923, but with technological advancements it was synthetically manufactured from recombinant DNA in 1977 and called “human insulin.”[10] There are two types of insulin: basal and mealtime. Basal insulins may be either intermediate or long-acting and help control insulin levels throughout the day. Mealtime insulins are either short- or rapid-acting and, as the name suggests, help control insulin levels around mealtime. Most people taking insulin, particularly those with Type 1 diabetes, use both a basal and mealtime insulin. There are also two types of active ingredients: human insulin and insulin analogs. Insulin analogs are modified in laboratories and typically provide better blood-sugar control than human insulin.[11] Also available is pre-mixed insulin, which combines either fast- and long-acting insulins or short- and intermediate-acting insulins, reducing the complexity and frequency of insulin administration.[12]
The improved blood sugar control provided by analog insulin led to a dramatic shift in patient use: In 2000, 96 percent of insulin users used human insulin and 19 percent used analog insulin; by 2010, only 15 percent of patients used human insulin while 92 percent used analog insulin.[13] Rapid-acting and ultra-long-acting analogs are now the most widely prescribed types of insulin.[14]
There are several other types of medications that people with Type 2 diabetes might take, besides insulin, such as those listed here.[15]
Insulin Administration
There are three primary ways to administer insulin, and the most common is through injection. There are now five types of injectable insulin that vary based on how quickly they start working and how long they continue working—rapid-acting, short-acting, intermediate-acting, long-acting, and ultra-long-acting.[16] Vials and syringes are the cheapest way to inject insulin and the most widely used. Insulin pens combine the vial and syringe in one product, providing greater dose accuracy and easier administration, but for that reason they are often more expensive than using a syringe.[17] Rapid-acting insulin has the most diverse administration options available, including syringe, pen, pump, or inhalation. Short-acting insulin can be administered through a syringe, pen, or pump, while intermediate, long, and ultra-long acting insulins can be administered only through a syringe or pen. Continuous subcutaneous insulin infusion (CSII) devices, commonly known as insulin pumps, are not manual injections but rather are battery-powered, computerized devices programmed to deliver short-acting insulin under the skin continuously throughout the day via a catheter attached to a person’s abdomen.[18] Insulin pumps are quite durable and can last for years at a time, and therefore they are generally more expensive than the supplies for manual injections.[19]
Another form of insulin administration is infusion, where insulin is injected directly into a vein through an IV. This is the preferred method to regulate blood glucose levels for diabetic patients in hospital settings, for example during surgery, labor and delivery, or in intensive care units.[20] The prevalence of IV insulin administration in hospitalized settings is unknown, but it is estimated that more than 20 percent of all adults discharged from hospitals have diabetes.[21]
Inhaled insulin is a relatively new form of insulin administration. Pfizer briefly introduced Exubera in 2006 but then voluntarily withdrew it from the market within its first year due to low sales.[22] In 2015, Mannkind Corporation marketed Afrezza in the United States. Inhaled insulin is rapid-acting and can be administered before meals to adults with type 1 or type 2 diabetes.[23] It is not a replacement for long-acting insulin and typically must be used in combination with injectable, long-acting insulin, meaning anyone using inhaled insulin is likely also using another type of insulin.
Patient Choice
There is a pronounced lack of choice for insulin regimens, and most of the choice that does exist is left up to a physician or medical provider. Physicians recommend treatment plans based on a person’s type of diabetes, lifestyle, and blood sugar levels throughout the day.[24] Some people only need one type of insulin, while others need multiple. For example, people with type 1 diabetes generally need injections of long-acting insulin once or twice a day, to mimic the body’s basal insulin, in addition to an injection of rapid-acting insulin to cover mealtime spikes in blood glucose levels. People with type 2 diabetes may only need long-acting insulin.[25] Patients are generally able to choose their preferred method of insulin administration, when the option is available, and they generally choose pens over syringes because they are more convenient, easier to administer, and more easily transportable.[26]
Chemical differences in insulin types and biological differences between diabetic patients make it so that there is very little competition in the insulin market. Patients may need to switch insulin plans for changes in blood sugar regulation, brand availability, or hormones and metabolism due to aging. Because most insulin products cannot substitute for each other, any changes must be prescribed or supervised by a doctor.[27] The variation between products and differences in individual blood sugar regulation make switching from one type of insulin to another dangerous, and potentially fatal, if not done correctly.[28] Furthermore, for some people, switching from one type or brand of insulin to another has caused allergic reactions with itching and swelling.[29] In short, insulin is not a normal commodity.
Nevertheless, cost considerations do come into play. Patients are more likely to follow and remain on a prescription plan correctly if it is affordable, and as a result physicians sometimes prescribe insulin plans that are the most cost-effective for a patient, but not necessarily the best for that patient’s medical needs.[30]
Insulin Patents and Market Exclusivity
Patents incentivize the creation of new processes and products through two pathways: securing the monetary gains from innovation by excluding others from using that invention for a fixed amount of time, and then democratizing information. In the United States, patent protections last for 20 years. Despite insulin being discovered nearly 100 years ago, many insulin products still benefit from patent protection. How can this be?
The insulin product discovered in 1921 and patented in 1923 was extracted from animals.[31] In 1978, insulin was, for the first time, reproduced from human recombinant DNA; this newly produced human insulin was patented and brought to market in 1982. Several new analog insulin products were developed between 1996 and 2005. The technological advances made over the past century have allowed for improved purity, enhanced concentration, increased supply, less crude extraction processes, easier administration, and varying lengths of effectiveness (reducing the number of injections needed daily) and quickness of onset, as discussed earlier.
Each time a company invents a new or improved way to produce or administer insulin or changes how long it lasts or how quickly it takes effect, it can receive a new patent. Thus, while insulin itself may no longer be on patent—or at least not the early insulin products brought to market—new insulin products (with, e.g., new non-active ingredients), manufacturing processes, or administration devices may be. As a result, there are currently hundreds of patents for insulin products.[32] A report from the World Health Organization (WHO) in 2016 found that by 2020, more than 90 percent of all existing patents for insulin products will have expired (excluding the patents on Pfizer’s inhaled insulin, which, as noted previously, was pulled from the market).[33] Other companies have filed new patent applications over the past several years, though, so the end of insulin patents is still at least 20 years away.
The seemingly constant stream of innovation does not mean, however, that patients have access to older, and therefore cheaper, but still sufficient insulin products. When a new product is brought to market, the older version is often either prescribed less frequently or possibly even discontinued by the manufacturer. Consequently, patients typically do not have access to many off-patent insulins. In some instances, the new version may significantly improve patient health and quality of life and is thus worth more to the patient, but sometimes the improvements are minimal, and the price increase may exceed the added value. WHO claims that not all inventions are, in fact, innovative, saying that medical innovation should be measured in terms of added clinical value and cost-effectiveness; not all newly patented insulin products meet this definition of innovation, WHO argues.[34] Conversely, Doctors Jeremy Greene and Kevin Riggs argue in the New England Journal of Medicine that “subsequent iterations of insulin represented actual innovations, each one being safer, more effective, or more convenient than its predecessor.”[35]
Besides patent protections, the Food and Drug Administration (FDA) separately grants new drugs periods of exclusivity of varying lengths—meaning the product is protected in some way from direct competitors. The length depends on the type of product and its approved uses. A drug may receive a period of exclusivity even if it does not receive a patent. Exclusivity refers to a period of time beginning upon the drug’s approval during which a product effectively has exclusive rights to be sold in the U.S. market. New biological products receive 4 years of filing exclusivity—during which would-be biosimilar manufacturers are prohibited from filing an application—and 12 years of approval exclusivity—during which the FDA is prohibited from approving a biosimilar application.
Biosimilar Insulin?
The nature of insulin itself has, to date, made it more difficult to generate effective market competition. Insulin is a biological product (“biologic”)—the first, in fact—though, when it was first approved by the FDA in 1982, it was technically designated simply as a “drug” for regulatory purposes under Section 505 of the Federal Food, Drug, and Cosmetic (FD&C) Act.[36] Biologics are produced from material from a living organism, not chemically synthesized molecules like most other manufactured drugs.[37] Thus, even once the patents and exclusivity periods expire, a competitor cannot make a generic version in the traditional sense. Biologic products, by their nature, cannot be identically duplicated, as is required for a product to be considered a generic; instead, a manufacturer may make a biosimilar product, whose molecular and biological structure shows no clinically meaningful difference from the biologic.
From a regulatory standpoint, however, there was not a legal pathway for biosimilars to come to market until the Affordable Care Act (ACA) created one; the Biologics Price Competition and Innovation Act included in the ACA created this pathway. Section 7002 of the ACA also included a provision officially changing insulin’s FDA designation (and all other biological products previously approved under Section 505 of the FD&C Act) from a “drug” to a biological product, as though it were approved under Section 351 of the Public Health Service Act—a change that then allows for biosimilars. This provision takes effect 10 years after the ACA’s enactment, allowing the first biosimilar insulin to be approved on March 23, 2020.[38] Section 605 of the 2019 year-end government funding bill also included a provision amending the definition of a biological product to now include any “chemically synthesized polypeptide.” The FDA argues this addition is important to ensure a biosimilar will also be able to come to market for any insulin that may be manufactured as a chemically synthesized polypeptide in the future.[39]
Thus, while the U.S. market has seen the introduction of a handful of follow-on insulin products (and biosimilar insulin products are available in Europe), there have not yet, technically, been any biosimilar insulin products in the U.S. market.
Biosimilars should lower the cost of insulin, but even with the new pathway competitors face challenges. The follow-on products introduced to date did not have a dramatic price-reducing effect, as was hoped, only reducing prices by about 10-15 percent.[40] (To be sure, this result may be largely due to the fact that even these follow-on products are manufactured by the three primary manufacturers rather than a competitor.[41]) Further, developing a biologic or a biosimilar is much more complex and much more expensive than producing small-chemical molecules, with the average cost an estimated 22 times greater for a biologic product.[42] This cost and complexity will likely slow the development of competitors in the insulin market and prevent the kinds of price drops that have occurred with small-molecule drugs.
Conclusion
Diabetes is one of the most burdensome diseases in America, and about 1.5 million individuals are newly diagnosed with diabetes each year. Soon, nearly 10 million Americans will need to take insulin every day to live. Three manufacturers dominate the current insulin market, and there is limited competition. That may soon change, however, as most patents for insulin products will have expired by the end of this year, and biosimilar insulin will finally be able to come to market. These factors may significantly change the market for insulin—and thus the lives of millions of Americans who depend on it.
[1] https://www.diabetes.org/resources/statistics/statistics-about-diabetes
[2] https://www.diabetes.org/resources/statistics/statistics-about-diabetes
[3] https://www.cdc.gov/media/releases/2017/p0718-diabetes-report.html
[4] https://ghr.nlm.nih.gov/condition/gestational-diabetes#definition
[5] https://care.diabetesjournals.org/content/41/6/1299
[6] https://care.diabetesjournals.org/content/41/6/1299
[7] https://www.hopkinsmedicine.org/news/media/releases/why_people_with_diabetes_cant_buy_generic_insulin
[8] https://fas.org/sgp/crs/misc/IF11026.pdf
[9] https://consumermedsafety.org/insulin-safety-center/item/418
[10] https://www.amjmed.com/article/S0002-9343(14)00585-3/pdf
[11] https://dtc.ucsf.edu/types-of-diabetes/type2/treatment-of-type-2-diabetes/medications-and-therapies/type-2-insulin-rx/types-of-insulin/insulin-analogs/
[12] https://dtc.ucsf.edu/types-of-diabetes/type2/treatment-of-type-2-diabetes/medications-and-therapies/type-2-insulin-rx/types-of-insulin/pre-mixed-insulin/
[13] https://jamanetwork.com/journals/jama/fullarticle/1878705
[14] https://www.amjmed.com/article/S0002-9343(14)00585-3/pdf
[15] https://www.healthline.com/health/diabetes/medications-list#type-2-diabetes
[16] https://www.diabetes.org/diabetes/medication-management/insulin-other-injectables/insulin-basics
[17] https://www.medicalnewstoday.com/articles/316607.php
[18] https://www.endocrineweb.com/guides/insulin/insulin-pump-overview
[19]https://dtc.ucsf.edu/types-of-diabetes/type2/treatment-of-type-2-diabetes/medications-and-therapies/type-2-insulin-rx/insulin-administration/
[20] https://dtc.ucsf.edu/types-of-diabetes/type2/treatment-of-type-2-diabetes/medications-and-therapies/type-2-insulin-rx/insulin-administration/
[21] https://www.ncbi.nlm.nih.gov/books/NBK279093/
[22] https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5001220/
[24] https://www.goodrx.com/blog/insulin-types-how-to-use/
[25] https://www.healthline.com/health/type-2-diabetes/basal-insulin/diabetes-statistics-and-basal-insulin-facts#3
[26] https://www.medicalnewstoday.com/articles/316607.php
[27] https://www.insider.com/safety-tips-for-switching-insulins-type-1-diabetes-patients-2019-8
[28] https://www.insider.com/safety-tips-for-switching-insulins-type-1-diabetes-patients-2019-8
[29] https://www.diabetes.co.uk/insulin/changing-insulin.html
[30]https://diabetescaucus-degette.house.gov/sites/diabetescaucus.house.gov/files/Congressional%20Diabetes%20Caucus%20Insulin%20Inquiry%20Whitepaper%20FINAL%20VERSION.pdf
[31] https://apps.who.int/medicinedocs/documents/s22481en/s22481en.pdf
[32] https://apps.who.int/medicinedocs/documents/s22481en/s22481en.pdf
[33] https://apps.who.int/medicinedocs/documents/s22481en/s22481en.pdf
[34] https://apps.who.int/medicinedocs/documents/s22481en/s22481en.pdf
[35] https://www.nejm.org/doi/pdf/10.1056/NEJMms1411398?articleTools=true
[36] https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4522877/
[37] https://www.biosimilarsresourcecenter.org/faq/what-are-biologics/
[38] http://housedocs.house.gov/energycommerce/ppacacon.pdf
[39] https://www.fda.gov/news-events/press-announcements/statement-low-cost-biosimilar-and-interchangeable-protein-products
[40] https://www.t1international.com/blog/2019/01/20/why-insulin-so-expensive/
[41] https://www.biosimilarsresourcecenter.org/faq/biosimilar-insulin-available/
[42] https://www.biosimilarsresourcecenter.org/faq/what-are-biologics/