Week in Regulation
January 3, 2023
Proposed Rules Finish Off 2022 With a Bang
The streak continues. The final week of 2022 marked yet another during which regulatory costs shot well past the billion-dollar threshold. There were 10 rulemakings with some measurable economic impact. The main items, however, included the Environmental Protection Agency’s (EPA) latest proposed rule on Renewable Fuel Standards for coming years and a Department of Health and Human Services (HHS) proposal that would update several aspects of Medicare. Across all rulemakings, agencies published $34.6 billion in total costs and added 3.6 million annual paperwork burden hours.
REGULATORY TOPLINES
- Proposed Rules: 25
- Final Rules: 60
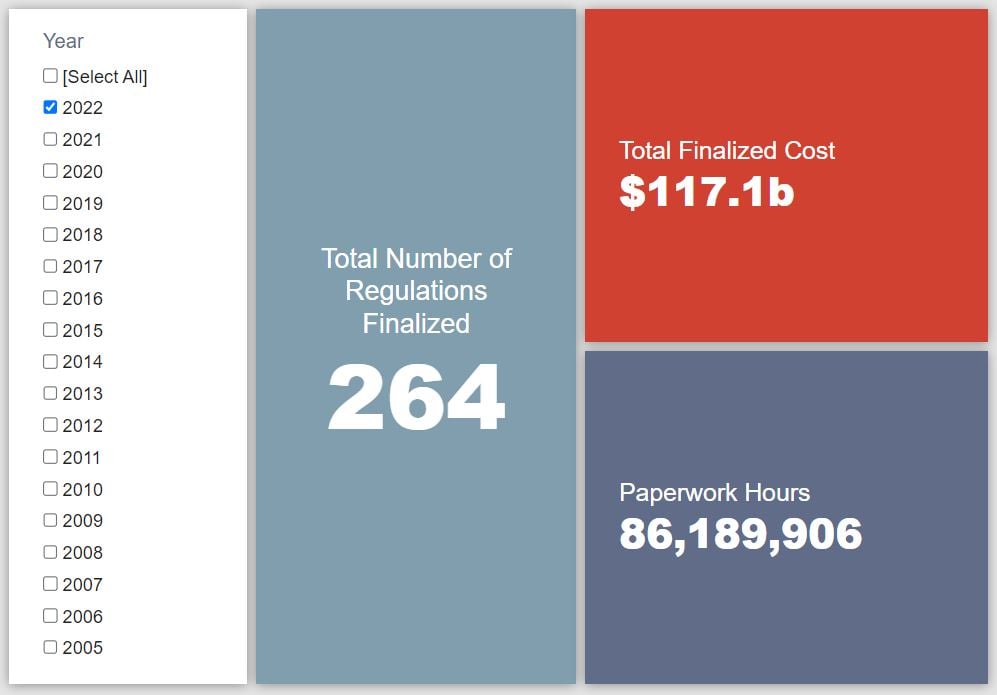
- 2022 Total Pages: 80,687
- 2022 Final Rule Costs: $117.1 billion
- 2022 Proposed Rule Costs: $191.2 billion
NOTABLE REGULATORY ACTIONS
The most consequential rulemaking of the week was the EPA proposed rule regarding “Renewable Fuel Standard (RFS) Program: Standards for 2023-2025 and Other Changes.” As the title suggests, this proposal represents EPA’s latest iteration of the RFS program established under the Energy Policy Act of 2005 and the Energy Independence and Security Act of 2007. In this latest edition, EPA is only able to provide monetized estimates for “Energy Security Benefits” and “Increased Societal Costs.” The former is a measure of the benefits of using domestic “renewable fuel” over imported fossil fuels, which EPA puts at $611 million. The latter, however, focuses on increased costs in the “production, blending, and distribution” of “renewable fuel” compared to fossil fuel and finds that it far outpaces the benefits figure, totaling nearly $28.5 billion across the three-year window the rulemaking covers.
The second-most notable rulemaking of the week was the HHS proposal covering a variety of Medicare programs. In particular, the rulemaking seeks:
To implement changes related to Star Ratings, medication therapy management, marketing and communications, health equity, provider directories, coverage criteria, prior authorization, passive enrollment, network adequacy, identification of overpayments, formulary changes, and other programmatic areas.
HHS expects these various changes to impose a wide array of new administrative burdens. As such, it estimates roughly 2.9 million hours of new paperwork annually and total costs of $5.8 billion over a 10-year period.
TRACKING THE ADMINISTRATIONS
As we have already seen from executive orders and memos, the Biden Administration will surely provide plenty of contrasts with the Trump Administration on the regulatory front. And while there is a general expectation that the current administration will seek to broadly restore Obama-esque regulatory actions, there will also be areas where it charts its own course. Since the AAF RegRodeo data extend back to 2005, it is possible to provide weekly updates on how the top-level trends of President Biden’s regulatory record track with those of his two most recent predecessors. The following table provides the cumulative totals of final rules containing some quantified economic impact from each administration through this point in their respective terms.
![]()
While this past week’s action tilted heavily to the proposed rule side, there were some modest shifts in the Biden Administration’s final rule costs and paperwork totals, increasing by $171 million and roughly 475,000 respectively. A Securities and Exchange Commission rule regarding “Insider Trading Arrangements and Related Disclosures” was the primary cause of such a shift. For the other two administrations, there were eerily similar changes in their cost totals with a $319 million increase under Trump and a $314 million increase under Obama. The “Bump Stock Rule” provided the bulk of the Trump-era increase. The Obama-era increase was more of a combined effort, however, with a nutrition labeling rule leading the way.
THIS WEEK’S REGULATORY PICTURE
This week, the Office of the Federal Register (OFR) says “stop the presses!” on the Public Papers of the Presidents of the United States (“Public Papers”).

https://www.archives.gov/federal-register/publications/presidential-papers.html
As the book closes on 2022, reference library aficionados will have fewer physical resources in the government documents section to pore over. OFR published a “final rule with request for comment” that discontinues the publication of the Public Papers as well as removing microfiche as an “official format” of Federal Register-related documents. The agency is undertaking this bit of administrative housekeeping based on the (likely well-founded) rationale that there is minimal public interest in such documents as they currently stand.
Dating back to 1957, the Public Papers were the semi-annual (generally released in six-month increments) compendiums of the Weekly Compilation of Presidential Documents, that then in turn became the Daily Compilation of Presidential Documents (“Daily Compilation”) in 2009. OFR finds that the public still has ample access to the Daily Compilation in each issue’s online edition going forward, making the publication of a collection of such records superfluous.
Furthermore, by the most recent estimate, the government sold “only approximately 13 to 20 copies of each volume” of the Public Papers in the past year. Given that level of interest, it is understandable that the government has decided to forgo its future production on either the online or hard-copy side. OFR also notes that digital versions of the Public Papers up through the Obama Administration will continue to remain available online.
The second main action in this rulemaking is the removal of “microfiche as an official format of the Federal Register and Code of Federal Regulations.” For those unfamiliar with this format, microfiche “is a format consisting of a flat sheet of film on which writing or other information is stored and which can be accessed using a microfiche reader.” It has historically been a way to preserve images of documents in a condensed format. The digitization of such documents, however, has now made it largely arcane and superfluous as well. For instance, out “of the more than 9,000 contractors…there is only one contractor that submitted bids for the creation of microfiche products in recent years.”
Overall, with this rulemaking, OFR is clearly catching up on its official policies to match practical reality.
TOTAL BURDENS
Since January 1, the federal government has published $308.3 billion in total net costs (with $117.1 billion in new costs from finalized rules) and 194.2 million hours of net annual paperwork burden increases (with 86.2 million hours in increases from final rules).