Weekly Checkup
July 24, 2020
It’s Back! The International Price Index Rears Its Ugly Head
Back in February, as part of his State of the Union address, President Trump was expected to announce policies aimed at reducing insulin prices and even moving forward with the long-touted International Price Index (IPI) proposal. The president was silent on drug prices, however, waiting instead for Congress to act legislatively. Now, with congressional action off the table, the president is expected to sign some number of executive orders today making good on many of his worst policy prescriptions for lowering drug prices. But whether any of the policies rumored to be announced today will ever be implemented is murky.
While details are scarce, the universe of policies under consideration include the truly terrible (the aforementioned IPI), the entirely pointless (drug importation), and the confusing (something simultaneously touching 340B hospitals, insulin, and EpiPen—don’t think too hard about it). Let’s look more closely at the three expected actions.
First, back in 2018, the administration issued an Advanced Notice of Proposed Rulemaking which would have tied Medicare Part B reimbursement for drugs to 126 percent of the average price paid by an index of countries, the IPI. American Action Forum experts have detailed the flaws here, but in effect the Trump Administration was proposing to import foreign price controls. This is the meat of what is anticipated this afternoon, but with one rumored change: Instead of paying 126 percent of the average price in the index, Medicare would pay no more than the lowest price paid by any country in the index. This is what is being referenced as “most favored nation.” The irony is that the administration has aggressively criticized a similar proposal from House Speaker Nancy Pelosi, saying it would lead to “as many as 100 fewer drugs entering the United States market over the next decade.” AAF experts have found that IPI as originally outlined could reduce new drug approvals by 30 new drugs over the same time period. The most substantive difference between Pelosi’s plan and Trump’s IPI is a matter of scale.
Second, in December the Trump Administration issued proposed rulemaking to allow for the importation of drugs from Canada. AAF experts have written extensively about why these proposed pathways won’t work; for a sampling read here and here. For one thing, Canada doesn’t have enough drugs to cover its own needs and sell to U.S. patients, and manufactures have every incentive to keep it that way. Additionally, Canadian pharmacies are unlikely to divert drugs to U.S. patients without a notable markup. Finally, the Canadian government itself has expressed opposition. Drug importation is a bipartisan idea that won’t die, but it also won’t work.
Finally there are rumors about a policy that will somehow leverage the 340B drug discount program (a whole mess in and of itself) to provide some limited population with low cost or even free insulin and EpiPens—one assumes within the hospital setting. We should get more details later today, but the expert consensus as of this morning seems to be: Huh?
What does all of this mean? The biggest and worst proposal, the IPI, still has a long way to go. Today’s executive order will likely direct the Department of Health and Human Services to move forward with proposed rulemaking. It’s unclear how quickly that can happen, but it’s possible the proposed rule is ready to go. Even if that is true, comment periods and other hoops make it unlikely a final rule could be promulgated before the election. The importation piece is further along, with a proposed rule already having been issued last year, but whether the administration can implement those policies before the election is somewhat irrelevant since, again, they won’t work. Finally, whatever has been cooked up on the insulin/EpiPen/340B front would at a minimum require a proposed rule and appears to be on a small enough scale as to be mostly inconsequential.
For now, these policies should be taken for what they are: campaign fodder. Whether there will be any interest in following through with them should the president be reelected remains to be seen.
Chart Review: Prisoner Releases and COVID-19 Infection Rates
Serena Gillian, Human Welfare Policy Fellow
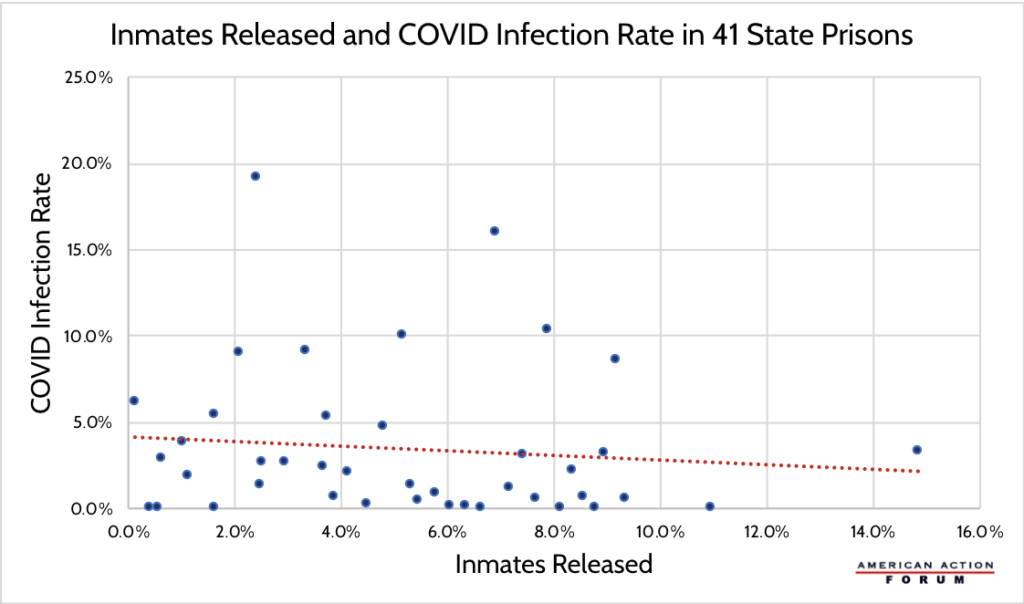
As COVID-19 cases reach new highs across the United States, inmates in prisons and jails are at a heightened risk of contracting the virus. In order to combat high infection rates, some correctional facilities are releasing inmates early to increase the potential for social distancing. The Marshall Project has been tracking new cases in state and federal prisons, and the Prison Policy Initiative has been collecting data on changes in prison and jail populations across the country. These data allow for an assessment of whether early release policies have had their desired impact. As the chart below shows, there is a correlation between higher rates of state prison inmates released and slightly lower COVID-19 infection rates. This limited effect points to a need for interventions beyond early release such as increases in personal protective equipment and comprehensive testing. It could also indicate that current reductions in state prison populations are not large enough to substantially reduce COVID-19 infections.
Worth a Look
New York Times: ‘It’s Like Groundhog Day’: Coronavirus Testing Labs Again Lack Key Supplies
Health Affairs: Designing Pull Funding For A COVID-19 Vaccine