Weekly Checkup
February 7, 2020
When, and Why, to Expect Administrative Action on Drug Prices
President Trump spent a lot of time talking about health care in his State of the Union speech, over 15 minutes in fact, but most significant might be what he didn’t say—and what it means for policy going forward.
The president touted—and perhaps overstated—his past administrative actions in relation to the individual market: short-term limited-duration plans, association health plans, health reimbursement arrangements, and hospital price transparency. He promised to protect people with preexisting conditions, and he hammered Democrats for their embrace of single-payer health care. But he didn’t propose any concrete steps on drug prices.
The lack of a concrete announcement is somewhat surprising. President Trump has made lowering prescription drug prices a pillar of his reelection campaign. In the lead-up to this week’s speech the administration signaled that the president could roll out plans for curtailing insulin costs in federal health programs and his long-discussed international price index (IPI) proposal—as previewed in last week’s Checkup. Instead of announcing a new and specific regulatory push on pharmaceutical prices, however, the president simply called on Congress to pass legislation to bring down prices and send it to him for his signature.
But President Trump may not have advanced the cause of drug legislation with his speech the way he hoped. While the president namechecked Senator Grassley, who has been pressing the case for his bipartisan drug price package in the Senate, the president did not give the sort of full-throated endorsement Grassley likely needs to bring more recalcitrant Senate Republicans on board his effort. Instead the speech left Congress largely in a stalemate around a number of different legislative efforts, none of which seems terribly viable at the moment.
By holding off on proposing the IPI rule, or other regulatory actions on drugs, the president left room for Congress to act before its self-imposed May deadline. In that sense, we’re back where we started the year, with a deadline, several proposals, and a shared desire on all sides to get something done, if not exactly the same thing. But in the aftermath of the speech, it feels like the air has gone out of the legislative effort anyway. Speaker Pelosi’s drug pricing legislation never had a chance in the Senate. House Democrats almost certainly won’t move forward with the House Republicans’ alternative bill. And without a strong, coordinated push from the White House, Senator Grassley won’t be able to convince Senate Republicans to swallow some his bill’s difficult policy pills that were necessary to bring Senate Democrats to the table in the first place.
As a result, a stay in administrative actions on drug pricing is likely only temporary. The legislative process will stumble along until May, but with the November election coming up, a late regulatory push still seems to be in the cards.
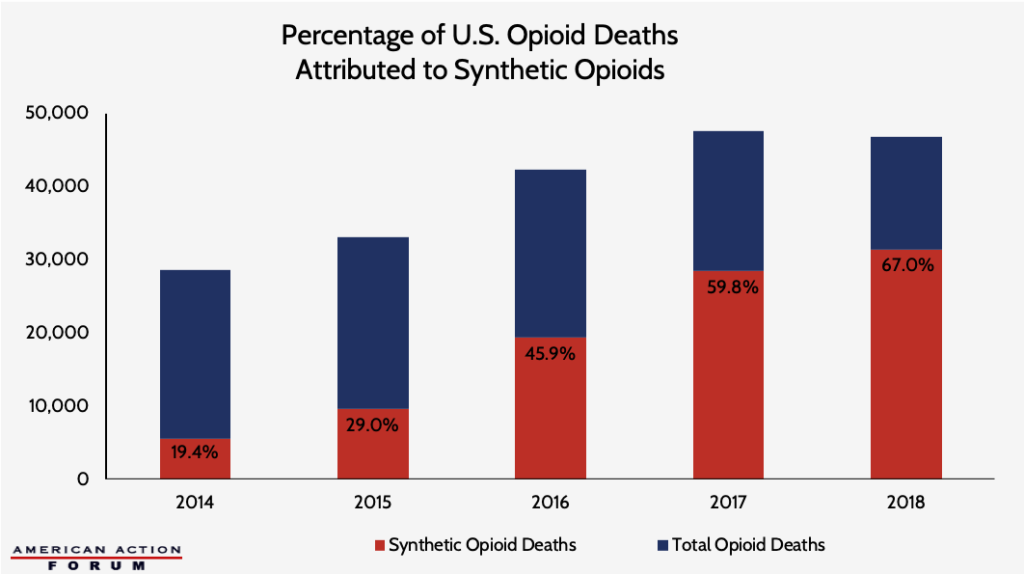
Chart Review: Breaking Down the Opioid-Overdose Decline
Josee Farmer, Health Care Policy Intern
U.S. life expectancy at birth increased in 2018, reversing the trend of the prior three years. The increase in life expectancy is due, in part, to the 4 percent decline in total drug overdose deaths from 2017 to 2018 – a total of 2,870 fewer deaths. Total opioid deaths, accounting for roughly 69 percent of overdose deaths in 2018, also decreased from 2017 to 2018. This decline, however, is entirely due to a drop in the number of deaths due to non-synthetic opioids. Synthetic opioid deaths—involving fentanyl, fentanyl analogs, and tramadol—were the only category of overdose deaths to increase from 2017 to 2018, a raw increase of 2,869 deaths. The percentage of U.S. opioid deaths attributed to synthetic opioids has steadily increased from 2014 to 2018, as the chart below demonstrates. Although total overdose deaths decreased from 2017 to 2018, the rise in synthetic opioid-related deaths indicates the challenge of opioid dependence remains.
Data obtained from the Center for Disease Control and Prevention
From Team Health
Testimony: More Cures for More Patients: Overcoming Pharmaceutical Barriers – Director of Human Welfare Policy Tara O’Neill Hayes
Restructuring Medicare Part D’s benefit could lower costs while not discouraging innovation, Hayes argued before the House Ways and Means Subcommittee on Health.
Worth a Look
Axios: Trump’s latest boost for Medicare Advantage
The Hill: House votes to condemn Trump Medicaid block grant policy