Insight
April 14, 2020
UPDATE: The Many Competing Proposals to Reform Medicare Part D
Executive Summary
Over the past year, Congress has put forward several proposals to lower prescription drug costs. This paper reviews some of these proposals.
- Three bills introduced in Congress, along with a recommendation from MedPAC, include plans to reform the Medicare Part D benefit structure that are similar to a proposal first put forward by the American Action Forum.
- Each of the plans seeks to put downward pressure on prescription drug prices by realigning incentives, specifically by requiring manufacturer rebates in (at least) the catastrophic phase of the benefit rather than (solely in) the coverage gap. Shifting the discount to this phase ensures the mandatory discount increases along with a drug’s price.
- Each proposal would also significantly increase insurer liability, increasing their incentive to control drug spending.
- Finally, each plan caps beneficiary spending and decreases significantly the federal government’s reinsurance liability in the catastrophic phase. These changes reduce the financial risk for both beneficiaries and taxpayers.
Introduction
Over the past year, Congress has put forward several proposals to lower prescription drug costs. One proposal that the Senate Finance Committee (Finance) has considered and approved is S. 2543, the Prescription Drug Pricing Reduction Act (PDPRA) of 2019. Among its several provisions, this legislation includes a redesign of the Medicare Part D Prescription Drug Program, similar to an American Action Forum (AAF) proposal from August 2018. The Senate Finance bill has since been modified, and this paper reflects those changes. Speaker Pelosi in September 2019 released another drug pricing reform package, H.R. 3, that also included an alternative proposal to redesign the Part D benefit structure. Then, in December 2019, the Republican leaders of the House Committees on Ways and Means, Energy and Commerce, and Judiciary introduced H.R. 19, the Lower Costs, More Cures Act; it too contains a proposal to restructure the Part D benefit. A companion bill with an identical Part D redesign proposal, S. 3129, was introduced in the Senate weeks later. Finally, the Medicare Payment Advisory Commission (MedPAC) announced recommendations for reforming the program’s benefit design in March 2020.
All of these proposals would be a significant departure from current law and would realign incentives for the various stakeholders in Medicare Part D. For an analysis of these proposals, click here.
Comparing the Proposals to Reform Medicare Part D
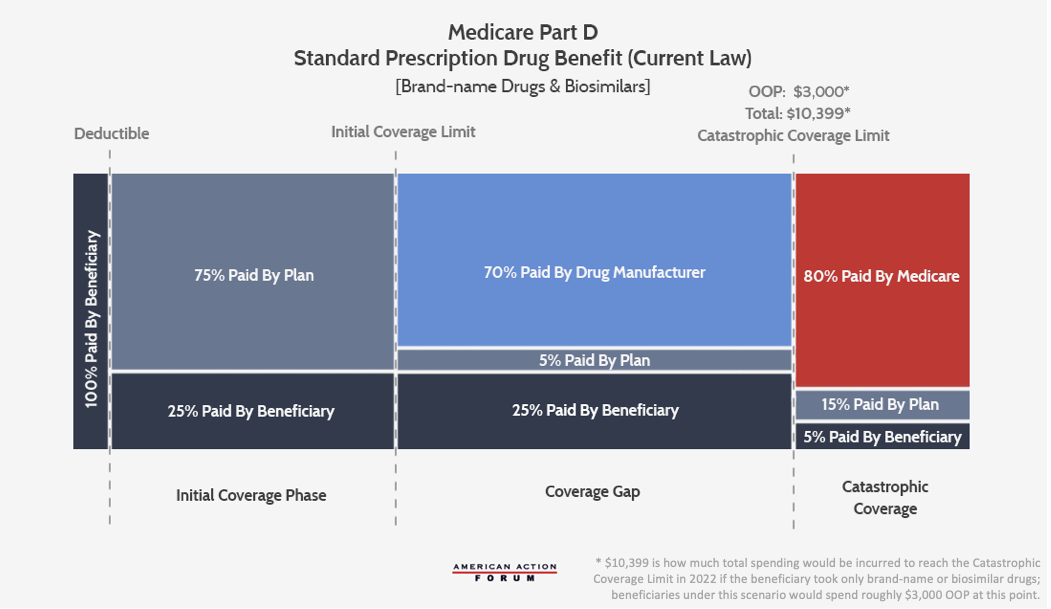
Current Law
The following graphic illustrates the current Part D benefit structure. Congress most recently reformed the program in 2018, and drug manufacturers now pay 70 percent of costs in the coverage gap while insurers pay just 5 percent during that phase. The federal government covers 80 percent of costs in the catastrophic phase and beneficiaries pay 5 percent of catastrophic costs, with no out-of-pocket (OOP) cap.
The current structure of Medicare Part D has several notable problems. Because insurer liability is now quite limited beyond the deductible, insurers have little incentive to keep beneficiaries out of the coverage gap and catastrophic phases. Further, drug manufacturers lack strong incentives to keep their prices down. The result of this lack of incentives is that the government is paying an increasing share of Part D’s cost in the catastrophic phase, and the government’s overall costs are rising. Finally, the current structure leaves some beneficiaries with very high costs even in the catastrophic phase. While these problems are significant, policymakers can restructure benefits to realign incentives and can employ market forces to put downward pressure on prices, as the AAF proposal illustrates.
AAF Proposal
The AAF proposal addresses the aforementioned problems by making several changes, shown below: It increases insurer liability in the catastrophic phase, moves the required manufacturer rebates to the catastrophic phase, and places a cap on beneficiaries’ OOP liability. The accounting firm Milliman modeled this plan and current law to find the parameters at which the expected manufacturer rebates under this proposal would be equal to what is expected under current law from 2020-2029. They estimated that an OOP cap of $2,500 would yield manufacturer rebates roughly equal to current law if manufacturers paid a 9 percent discount in the catastrophic phase for all beneficiaries.[1] Following MedPAC’s lead, the AAF proposal sets the government’s reinsurance liability at 20 percent, leaving insurers liable for 71 percent of costs in the catastrophic phase.
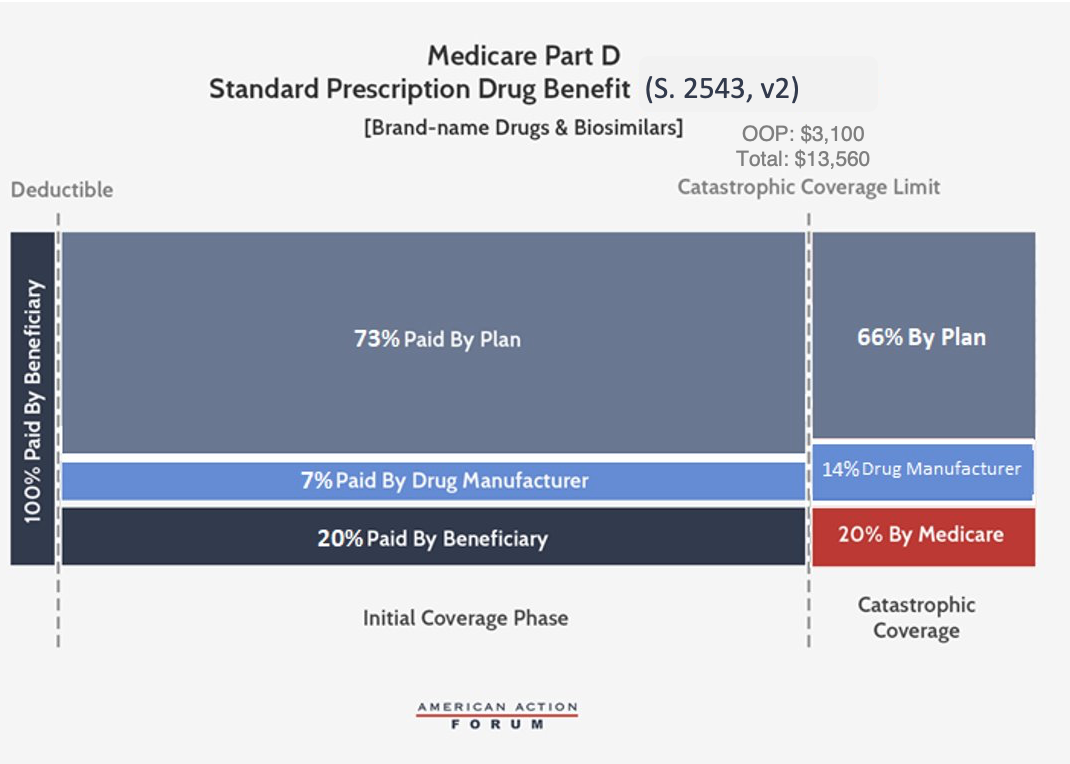
Senate Finance Proposal, S. 2543 (revised)
The Senate Finance Committee’s (Finance) Prescription Drug Pricing Reduction Act (PDPRA) of 2019 originally included a similar conceptual framework to the AAF proposal, but Finance is now working on a revised plan and many of the details have been made public; the proposal discussed here reflects those changes. The revised Finance proposal, which would be phased-in beginning in 2022, sets an OOP limit of $3,100, equal to roughly the amount of OOP spending before the catastrophic threshold expected under current law. Beneficiary coinsurance would also be reduced to 20 percent for brand-name drugs and biosimilars. The proposal would require brand-name and biosimilar drug manufacturers to pay a 14 percent rebate for all drug costs incurred after the beneficiary reaches the catastrophic phase. Further, manufacturers must pay 7 percent of costs during the initial coverage phase, as well, meaning rebates will be required for all drugs taken after a beneficiary reaches the deductible. Insurer liability in the initial coverage phase would be 73 percent for brand-name drugs and biosimilars, and in the catastrophic phase it would gradually increase to 66 percent for such drugs by 2024. When a patient uses a generic drug, the insurer’s liability would increase to cover the share not paid by the drug manufacturer. Federal reinsurance would be reduced to 20 percent for all drugs by 2024. Finally, the revised Finance legislation would also reduce beneficiaries’ premium liability from 25.5 percent to 24.5 percent.
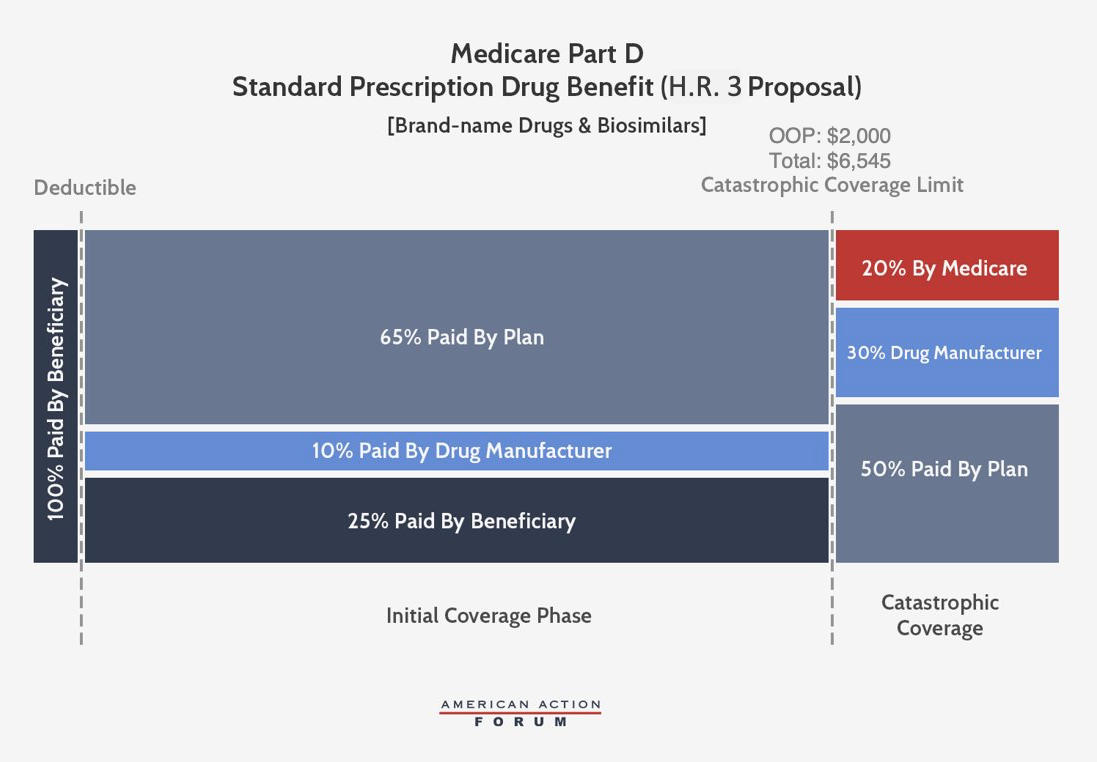
Speaker Pelosi’s Proposal, H.R. 3
Speaker Pelosi’s proposal is similar to the revised Finance proposal but sets different parameters that extract even more money from manufacturers. This plan would cap beneficiary OOP spending at $2,000—about $600 less than what an average beneficiary reaching catastrophic coverage pays in 2019. Brand-name and biosimilar drug manufacturers would be required to pay 10 percent of costs in the initial coverage phase and 30 percent of costs incurred after the beneficiary reaches the catastrophic phase—more than triple the levels needed to replicate manufacturer liability under the current structure. Insurer liability in the initial coverage phase would thereby be reduced to 65 percent when a beneficiary takes a brand-name drug or biosimilar. In the catastrophic phase, insurer liability would increase to 50 percent for brand-name drugs and biosimilars and 80 percent for generic drugs, while federal reinsurance would cover the remaining 20 percent.
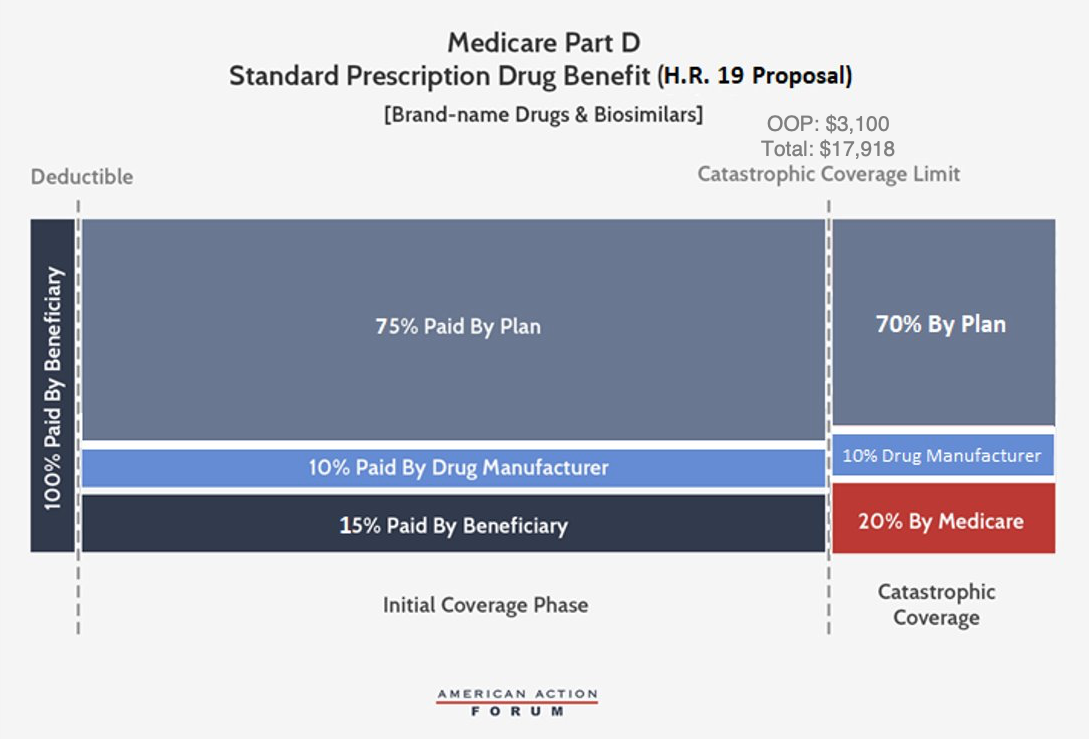
House Republicans’ Proposal, H.R. 19, and Senate Companion, S. 3129
H.R. 19 and S. 3129, like the two other congressional proposals, would require manufacturer discounts in both the initial coverage phase and the catastrophic phase, with a steady liability of 10 percent of the costs above the deductible. As in the Finance proposal, these bills would set the OOP limit at $3,100; beneficiary coinsurance for brand-name drugs and biosimilars would be reduced to 15 percent, while remaining at 25 percent for generics. Insurers would be responsible for 75 percent of all costs in the initial coverage phase and 70 percent in the catastrophic phase, regardless of the type of drug taken. Government reinsurance would decrease to 20 percent when a manufacturer owes a discount and to 30 percent when it does not.
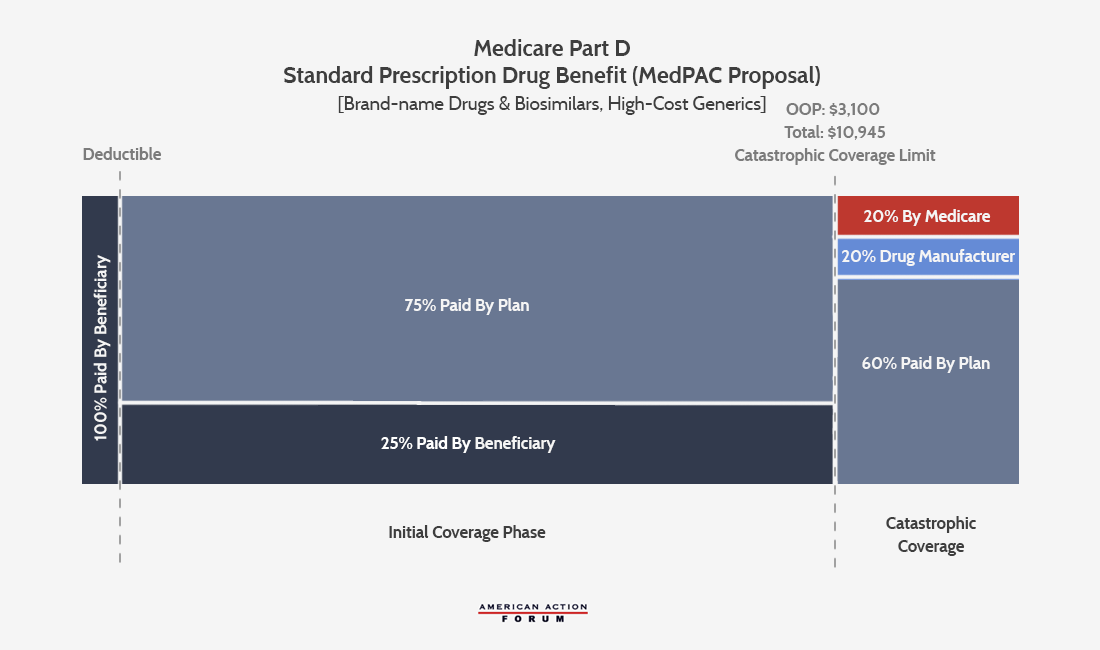
MedPAC’s Recommendation
MedPAC’s proposal, as outlined in March 2020, is more similar to the AAF proposal in that it only requires manufacturer discounts in the catastrophic phase. The proposal would set the OOP cap at $3,100, and beneficiary coinsurance would remain at 25 percent. Insurer liability in the initial coverage phase would cover the remaining 75 percent. In the catastrophic phase, manufacturers of brand-name drugs, biosimilars, and high-priced generics would be responsible for 20 percent of the costs. Insurers would be responsible for 60 percent of the costs of such drugs and 80 percent when a beneficiary takes a lower-price generic. MedPAC’s proposal would reduce federal reinsurance to 20 percent for all products in the catastrophic phase.
MedPAC also recommends increasing copayment amounts for beneficiaries eligible for the low-income subsidy (LIS) enrollees taking nonpreferred and nonformulary drugs; giving plans greater flexibility to manage use of drugs in the protected classes; and allowing plans to establish preferred and nonpreferred specialty tiers. MedPAC also suggests recalibrating the program’s risk-adjustment model to account for plans’ increased liability and temporarily modifying the risk corridor parameters during the transition to the new benefit design.
Comparing the Proposals
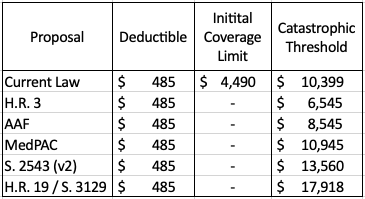
In summary, each proposal would eliminate the coverage gap that currently exists, reducing the number of coverage phases to three, as shown in Table 1. The variation in how much total drug spending must occur before reaching the catastrophic threshold depends on the beneficiaries’ OOP limit as well as their coinsurance rate before that limit.
Table 1: Coverage Phase Thresholds, 2022
Key Reform Component: The Maximum Manufacturer Liability Per Drug
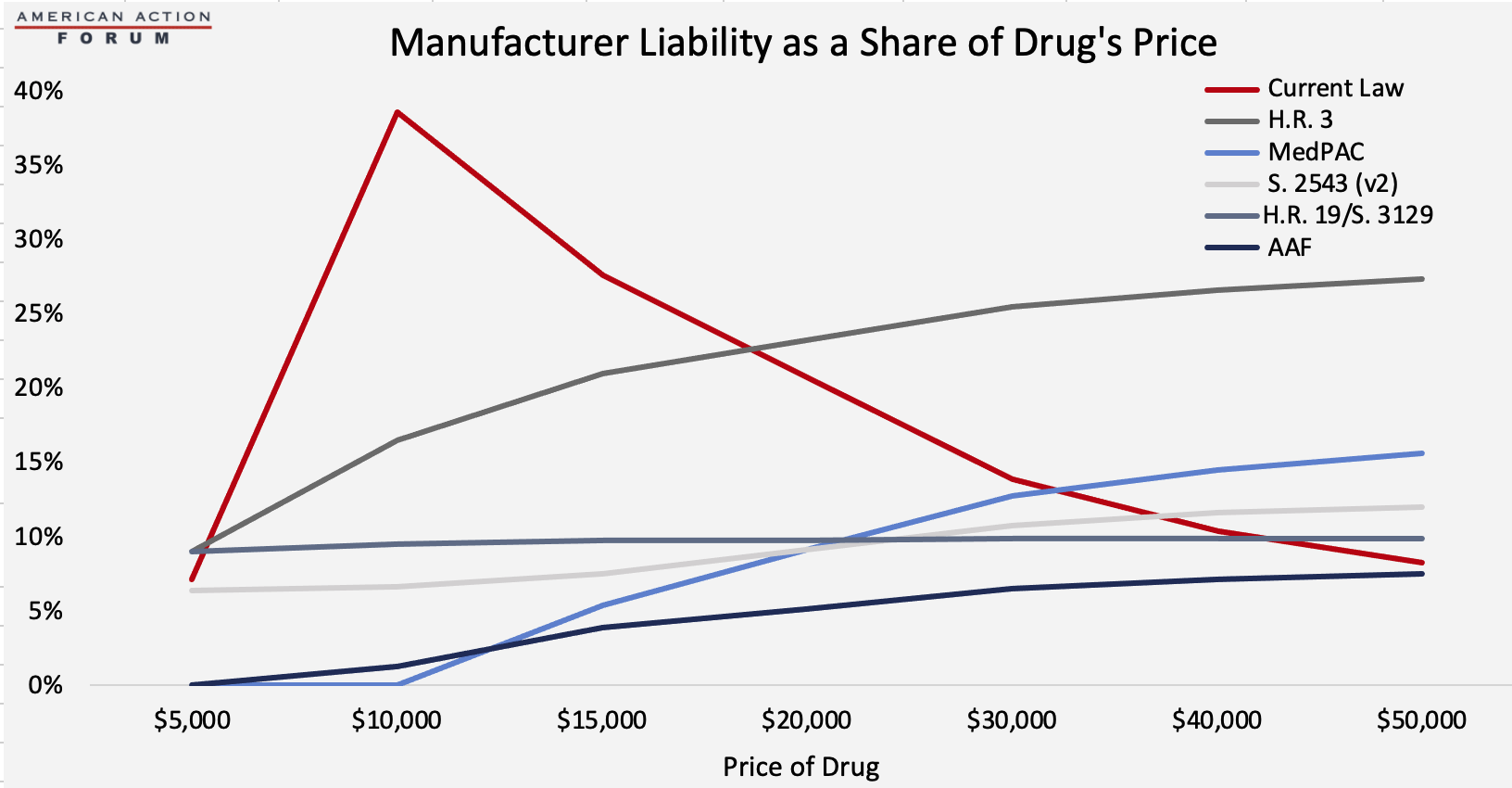
Under each proposal, the required rebate amount for a given drug can be calculated based on the drug’s price. Under current law, manufacturers only have to pay a rebate in the coverage gap, and as a result, the coverage gap rebate shrinks proportionately as the price of the drug increases because the rebate amount is capped indirectly. For example, a drug costing $9,303 in 2020 (the lowest cost at which the maximum rebate must be paid) pays a rebate of $3,698, equal to nearly 40 percent of the cost of the drug, as explained here. A drug priced at $50,000, on the other hand, pays the same rebate amount, but that amount is equal to only 8 percent of the drug’s price. Thus, the current benefit design is more harmful to lower-priced drugs, as shown by the early peak of the red line in the chart below contrasted with the gradual increase of the other lines. If the program’s benefit design required the rebate in the catastrophic phase of coverage, however, the rebate would no longer be capped, and the required rebate amount would increase, both nominally and as a share of the drug’s price, as the price increases, as shown under each of the proposals. As a result, more of the burden of the rebates would be placed on higher-priced drugs.
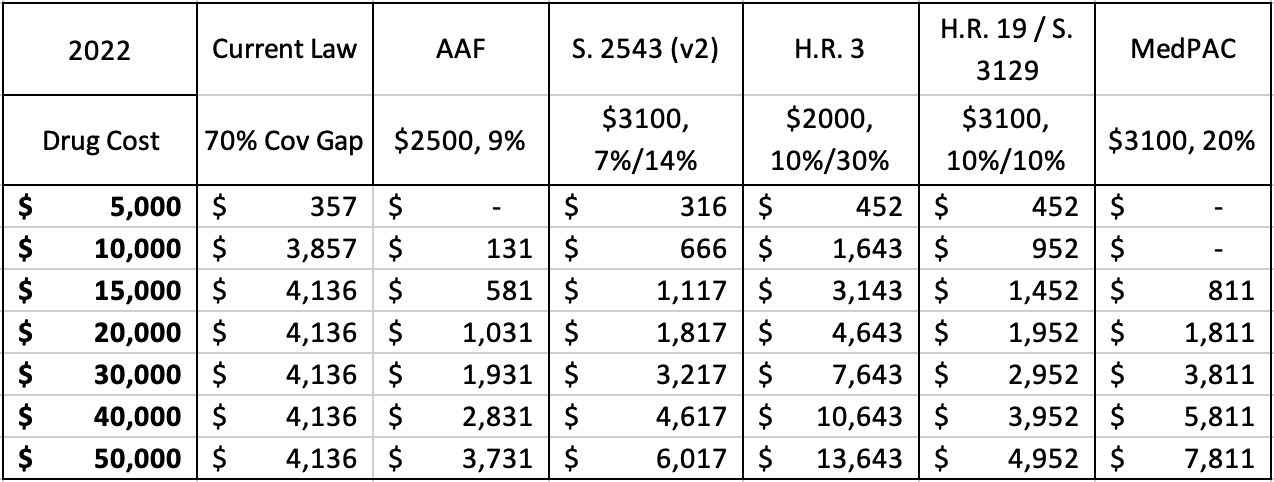
Table 2: Maximum Manufacturer Liability Under Each Proposal by Drug’s Price, 2022
Conclusion
Each of the proposals for reforming the Medicare Part D benefit structure contain four key components: capping beneficiary OOP spending, increasing insurer liability, requiring manufacturer liability to increase along with a drug’s price, and decreasing the government’s reinsurance liability. These changes should provide beneficiaries and taxpayers with less financial risk and increase insurers’ and drug manufacturer’s incentive to keep drug costs down. Nevertheless, while the plans are each conceptually similar, the differences in the details will result in significant variations in their impact, as detailed here.
[1] Current law does not require manufacturers to pay Coverage Gap discounts for low-income subsidy (LIS) beneficiaries.