Insight
June 11, 2019
The Impact of Shifting Rebates to Catastrophic Coverage in Medicare Part D
Executive Summary
- Congress is considering reforms to the Medicare Part D program that include moving the current Coverage Gap Discount Program (CGDP) to the catastrophic phase of coverage.
- Under current law, the maximum rebate amount required of manufacturers for a given drug in 2020 is $3,698, regardless of the drug’s cost, and the majority of mandatory rebates are paid for drugs that cost $480-$580.
- If manufacturers were instead required to pay a rebate in the catastrophic phase, a much greater share of the rebates paid would be for the most expensive drugs.
- This reform would result in the mandatory rebate amount increasing with the drug’s price, which would provide an incentive against price inflation—an incentive the current system lacks.
Introduction
In the 16 years since Congress created the Medicare Part D program, the prescription drug market, insurance structure, and pricing practices have changed. Patterns have emerged that make it clear the current system is encouraging undesirable behaviors that increase costs for the government and consumers. Reforming the benefit structure in a way that realigns the financial incentives of both the insurers and drug manufacturers may help to reverse these trends by putting downward pressure on drug prices.
There are currently four phases of coverage: a first, where the beneficiary covers all costs up to the deductible; a second, the initial coverage phase; a third (the “coverage gap”), when the manufacturer now covers 70 percent of costs; and a fourth, the catastrophic phase, when the government covers 80 percent of costs and the insurer 15 percent. The beneficiary is responsible for some costs in all four phases.
Congress is considering providing beneficiaries with an out-of-pocket (OOP) cap and reducing the government’s share of the costs in the catastrophic phase while increasing insurer’s liability. These changes are a good start, but alone they are insufficient to provide significant relief for beneficiaries or put meaningful downward pressure on drug prices. Prior work by the American Action Forum details why this is so and proposes a more comprehensive reform of the benefit structure, including moving the mandatory drug manufacturer rebates from the coverage gap to the catastrophic phase, as shown below.
This paper provides new analysis of the potential impacts of such a reform. It looks at the rebates that would result from requiring rebates at various rates from drugs at different prices.
The Proposed Reform
The current CGDP limits the mandatory rebate amount because the coverage gap only covers a limited amount of expenditures. In 2020, the most that a drug manufacturer will be required to pay in coverage gap rebates for a single drug, now that the mandatory rebate is 70 percent, is $3,698.[1] Any drug costing $9,303 or more will require a payment of this amount and no more. Accordingly, the existing structure does not provide any incentive to drug manufacturers to limit prices once they’ve exceeded the catastrophic threshold. By requiring the discount instead to be paid in the catastrophic phase, which has no limit other than the end of the year, the mandatory rebate will increase with the price of the drug. This change should—at least more than the current system—discourage price growth.
Further, this change will target the rebate toward costlier drugs. MedPAC found that under the current CGDP, diabetic therapies are responsible for 31 percent of rebates paid; asthma and chronic obstructive pulmonary disease (COPD) drugs are responsible for 12 percent, and 11 percent are paid for anti-coagulants.[2] The average price of these medicines is $480-$580 per claim.[3] Alternatively, if the rebates were collected in the catastrophic phase, MedPAC estimates that antineoplastics (chemotherapy) would account for 20 percent of the rebates; antivirals would be responsible for 15 percent; diabetic therapies, 11 percent; analgesics and anti-inflammatory drugs would account for 9 percent; central nervous system agents another 9 percent; and Multiple Sclerosis drugs would account for 8 percent.[4] The price of these drugs ranges from a few thousand to more than $30,000.[5]
If the OOP limit (which would trigger a move into the catastrophic phase) was set at $2,500, the total amount of drug expenditures that would be incurred by all parties at that point would equal $8,695 in 2020. For comparison, under current law, beneficiaries themselves are expected to spend $2,652 OOP before reaching catastrophic coverage in 2020—with a True OOP (TrOOP) of $6,350—and total drug expenditures will equal $9,303, if the beneficiary is only taking brand-name drugs.[6]
Understanding Different Rebate Rates
According to analysis by actuarial accounting firm Milliman, 9 percent was estimated to be the rebate discount rate in the catastrophic phase at which overall manufacturer rebates would be roughly equal to what they are expected to be under current law.[7]
This figure provides a useful starting point for analyzing various discount rates. If the manufacturer rebate were moved to the catastrophic phase and set at a rate of 9 percent with a catastrophic threshold limit of $2,500, the break-even price, at which point the rebate would be equal to the amount currently required in 2020, is $49,784. Every manufacturer with a drug costing more than this will pay more under these parameters than under the current system. Further, every manufacturer with a drug costing more than $8,695 will pay something, though it will be less than what they pay now if the cost is below that break-even price. (Technically, all drugs provided to beneficiaries after they’ve reached the catastrophic phase will require the payment of a rebate, regardless of price, but it is impossible to know what those drugs might be, if their cost is less than $8,695, and it may simply be poor timing that they become subject to the rebate requirement.)
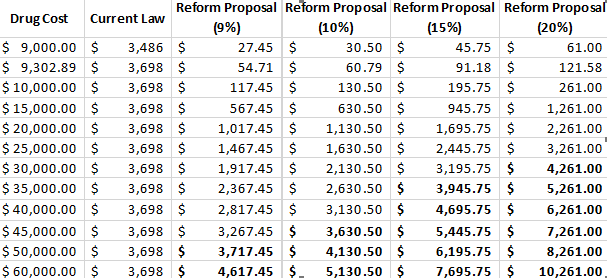
Of course, even if overall rebates are the same, some manufacturers would fare better and some would fare worse, depending on the prices of their drugs and their utilization rates. Considering prices, rather than annualized costs, is relevant here because there are many drugs for which a single dispense will push beneficiaries into the catastrophic phase. In 2017, there were 152 drugs for which a single claim exceeded the catastrophic threshold. There were nearly 2 million claims for these drugs, resulting in total spending of $24.6 billion, about a quarter of overall spending in Medicare Part D. The table below shows the various rebate amounts that are required for drugs at different price points under current law compared with the rebates required under various discount rates if the rebate were moved to the catastrophic phase with an OOP threshold of $2,500 (all based on a standard benefit design in 2020).
The bolded numbers in the table highlight costs over (or nearly at) the break-even price points. As previously stated, if the discount rate is set at 9 percent in the catastrophic phase, the break-even price is $49,784. If the discount rate is set at 10 percent, the break-even price drops to $45,675. At 15 percent, the break-even price is $33,348; and at 20 percent, it is $27,185.
According to the Centers for Medicare and Medicaid Services (CMS), there were 361 drugs in 2017 for which average spending per beneficiary for the year exceeded $8,695, and thus would be required to pay some rebate amount if the mandatory rebate were moved to the catastrophic phase with a threshold of $2,500.[8] These drugs were provided to more than 1.5 million beneficiaries at a total cost of $49.9 billion. An estimated $36.4 billion would have been spent on these drugs in the catastrophic phase at that threshold. Assuming rebates were required for all beneficiaries—not just non-low-income subsidy (LIS) beneficiaries as under current law—a 9 percent rebate on just these drugs would provide $3.3 billion in mandatory rebates (though this figure does not account for the enrollment and price growth that has occurred since 2017, nor the drugs that cost less than $8,695 but will inadvertently be required to pay a rebate simply because they’re being provided after the beneficiary has reached catastrophic coverage). If the discount rate was set at 10 percent, the rebates would total $3.6 billion. At 15 percent, the rebates would equal $5.5 billion, and a 20 percent rate would yield $7.3 billion (again, not accounting for growth in enrollment, which is expected to increase 10 percent between 2017 and 2020, or growth in prices since 2017). It is estimated that coverage gap rebates totaled nearly $6 billion in 2017.[9]
If the prices for the drugs covered in 2017 increased 5 percent between 2017 and 2020 and the number of beneficiaries increased by 10 percent (the rate that overall enrollment is expected to have increased during this period), then an additional 17 drugs provided to an additional 216,000 beneficiaries would be subject to the rebate. The total estimated spending for these 378 drugs based on these assumed growth rates would equal $58.2 billion, of which roughly $42.8 billion would occur in the catastrophic phase. A 9 percent discount rate would yield $3.9 billion in rebates for these drugs. A 10 percent discount rate, $4.3 billion; 15 percent, $6.4 billion; and 20 percent, $8.6 billion. These figures do not account for any new drugs that have or will come to market since 2017 and that may exceed the threshold.
Milliman also estimated that if such reforms led to reductions in brand-name price growth of 5 percent, beneficiaries and the federal government would see overall savings of $31 billion over 10 years.[10]
Potential Benefits of Reform
The benefit structure described here would provide much-needed financial protection for beneficiaries with high medication costs. The realignment of the liabilities in the catastrophic phase would encourage both insurers and drug manufacturers to keep costs down while protecting taxpayers from rising reinsurance costs. Finally, this change will target the program’s benefits and mandatory drug rebates more appropriately than the current structure.
[1] In 2020, a beneficiary will reach the coverage gap after $4,020 in total drug expenditures, of which the beneficiary will have paid $1,390 in OOP expenses. The beneficiary will exit the coverage gap after $6,350 in TrOOP costs, $2,652 of which will be paid by the beneficiary and $3,698 of which will be paid by drug manufacturers if the beneficiary is taking only brand-name drugs. If the beneficiary is taking just one drug, then this is the maximum amount a manufacturer would have to pay for a given drug per beneficiary. (Calculations my own. 2020 plan information found here: https://q1medicare.com/PartD-The-2020-Medicare-Part-D-Outlook.php)
[2] Schmidt, Rachel and Shinobu Suzuki, MedPAC. “Options to increase the affordability of specialty drugs and biologics in Medicare Part D.” April 5, 2019. http://www.medpac.gov/docs/default-source/default-document-library/options-to-increase-the-affordability-of-specialty-drugs-in-pt-d—final.pdf?sfvrsn=0 (Slide 9)
[3] Schmidt, Rachel and Shinobu Suzuki, MedPAC. “Options to increase the affordability of specialty drugs and biologics in Medicare Part D.” April 5, 2019. http://www.medpac.gov/docs/default-source/default-document-library/options-to-increase-the-affordability-of-specialty-drugs-in-pt-d—final.pdf?sfvrsn=0 (Slide 9)
[4] Schmidt, Rachel and Shinobu Suzuki, MedPAC. “Options to increase the affordability of specialty drugs and biologics in Medicare Part D.” April 5, 2019. http://www.medpac.gov/docs/default-source/default-document-library/options-to-increase-the-affordability-of-specialty-drugs-in-pt-d—final.pdf?sfvrsn=0 (Slide 13)
[5] Schmidt, Rachel and Shinobu Suzuki, MedPAC. “Options to increase the affordability of specialty drugs and biologics in Medicare Part D.” April 5, 2019. http://www.medpac.gov/docs/default-source/default-document-library/options-to-increase-the-affordability-of-specialty-drugs-in-pt-d—final.pdf?sfvrsn=0 (Slide 13)
[6] Note that TrOOP includes the rebate amounts provided by manufacturers. If the manufacturer rebates are moved from the coverage gap to the catastrophic phase, manufacturer rebates would no longer count toward a beneficiary’s TrOOP. That is why a lower OOP threshold, relative to the current TrOOP threshold, is needed to achieve a similar level of OOP spending directly by the beneficiary.
[7] Based on Milliman’s estimate of rebate revenues from 2020-2029, under current law, assuming no behavioral changes. http://us.milliman.com/uploadedFiles/insight/2018/restructuring-Medicare-Part-D-benefit.pdf
[8] Based on average annual spending per beneficiary, according to the CMS Medicare Part D Drug Spending Dashboard. https://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-and-Reports/Information-on-Prescription-Drugs/MedicarePartD.html
[9] https://www.americanactionforum.org/insight/understanding-the-policies-that-influence-the-cost-of-drugs/#_ednref11
[10] http://us.milliman.com/uploadedFiles/insight/2018/restructuring-Medicare-Part-D-benefit.pdf